Beyond Needles: Immunomodulatory Hydrogel-Guided Vaccine Delivery Systems
- PMID: 39851978
- PMCID: PMC11764567
- DOI: 10.3390/gels11010007
Beyond Needles: Immunomodulatory Hydrogel-Guided Vaccine Delivery Systems
Abstract
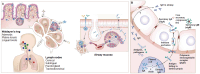
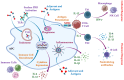
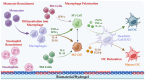
Vaccines are critical for combating infectious diseases, saving millions of lives worldwide each year. Effective immunization requires precise vaccine delivery to ensure proper antigen transport and robust immune activation. Traditional vaccine delivery systems, however, face significant challenges, including low immunogenicity and undesirable inflammatory reactions, limiting their efficiency. Encapsulating or binding vaccines within biomaterials has emerged as a promising strategy to overcome these limitations. Among biomaterials, hydrogels have gained considerable attention for their biocompatibility, ability to interact with biological systems, and potential to modulate immune responses. Hydrogels offer a materials science-driven approach for targeted vaccine delivery, addressing the shortcomings of conventional methods while enhancing vaccine efficacy. This review examines the potential of hydrogel-based systems to improve immunogenicity and explores their dual role as immunomodulatory adjuvants. Innovative delivery methods, such as microneedles, patches, and inhalable systems, are discussed as minimally invasive alternatives to traditional administration routes. Additionally, this review addresses critical challenges, including safety, scalability, and regulatory considerations, offering insights into hydrogel-guided strategies for eliciting targeted immune responses and advancing global immunization efforts.
Keywords: adjuvants; biomaterials; hydrogels; immunogenicity; immunomodulation; vaccine delivery.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
-
- Priyanka, Abusalah M.A.H., Chopra H., Sharma A., Mustafa S.A., Choudhary O.P., Sharma M., Dhawan M., Khosla R., Loshali A., et al. Nanovaccines: A Game Changing Approach in the Fight against Infectious Diseases. Biomed. Pharmacother. 2023;167:115597. doi: 10.1016/j.biopha.2023.115597. - DOI - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

