Suction tube uterine tamponade versus uterine balloon tamponade for treatment of refractory postpartum hemorrhage: A randomized clinical feasibility trial
- PMID: 39853765
- PMCID: PMC12177279
- DOI: 10.1002/ijgo.16164
Suction tube uterine tamponade versus uterine balloon tamponade for treatment of refractory postpartum hemorrhage: A randomized clinical feasibility trial
Abstract
Objective: To compare low-cost "Suction Tube Uterine Tamponade" (STUT) treatment for refractory postpartum hemorrhage (PPH) with uterine balloon tamponade (UBT) using a randomized feasibility study.
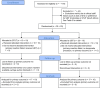
Methods: After verbal assent, we allocated participants with refractory PPH by randomly ordered envelopes to STUT or routine UBT at 10 hospitals in South Africa and one tertiary referral center in Colombia between January 10, 2020, and May 3, 2024. In the STUT group, we inserted a 24 FG Levin stomach tube into the uterine cavity and applied suction. The control group received standard UBT, mainly the Elavi free-flow balloon or the Bakri fixed volume balloon. There were fundamental differences between the South African and the Colombian sites, so the pre-specified analysis combined data from the two countries by meta-analysis.
Results: We enrolled 59 participants. The rate of the primary outcome (blood loss >1000 mL or laparotomy or death) was 8/27 (30%) in the STUT group versus 14/27 (52%) in the UBT group (risk ratio [RR] 0.56, 95% confidence interval [CI] 0.30-1.05, P = 0.07). Per protocol analysis was 7/26 (27%) versus 15/28 (54%) (RR 0.49, 95% CI 0.25-0.96, P = 0.04). Reporting severe pain during the procedures was less frequent in the STUT group (RR 0.46, 95% CI 0.25-0.86, P = 0.01). Most secondary outcomes favored the STUT group, with low certainty.
Conclusions: STUT was experienced as less painful than UBT. Results were consistent with reported observational findings and one other randomized trial evidence of greater effectiveness for suction than balloon tamponade.
Keywords: feasibility; postpartum hemorrhage; randomized clinical trial; suction tube uterine tamponade; uterine balloon tamponade.
© 2025 The Author(s). International Journal of Gynecology & Obstetrics published by John Wiley & Sons Ltd on behalf of International Federation of Gynecology and Obstetrics.
Conflict of interest statement
GJH has received consultancies as inventor of the Maternawell calibrated tray for blood loss monitoring after birth, which was not used in this study. The other authors have no conflicts of interest.
Figures

References
-
- Gallos I, Devall A, Martin J, et al. Randomized trial of early detection and treatment of postpartum hemorrhage. N Engl J Med. 2023;389(1):11‐21. - PubMed
-
- Revert M, Cottenet J, Raynal P, Cibot E, Quantin C, Rozenberg P, Intrauterine balloon tamponade for management of severe postpartum haemorrhage in a perinatal network: a prospective cohort study, BJOG 2017;124(8):1255–1262 - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

