Ischemic Myocardium Targeting Peptide-Guided Nanobubbles for Multimodal Imaging and Treatment of Coronary Microvascular Dysfunction
- PMID: 39853877
- PMCID: PMC12083440
- DOI: 10.1002/adhm.202404477
Ischemic Myocardium Targeting Peptide-Guided Nanobubbles for Multimodal Imaging and Treatment of Coronary Microvascular Dysfunction
Abstract
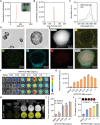
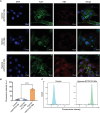
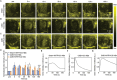
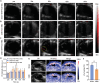
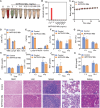
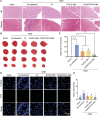
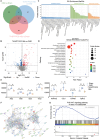
Coronary microvascular dysfunction (CMD) refers to clinical symptoms caused by structural and functional damage to coronary microcirculation. The timely and precise diagnosis of CMD-related myocardial ischemia is essential for improving patient prognosis. This study describes a method for the multimodal (fluorescence, ultrasonic, and photoacoustic) noninvasive imaging and treatment of CMD based on ischemic myocardium-targeting peptide (IMTP)-guided nanobubbles functionalized with indocyanine green (IMTP/ICG NBs) and characterizes their basic characteristics and in vitro imaging and targeting abilities. The IMTP/ICG NBs enable the accurate location of myocardial ischemia via photoacoustic imaging, and when loaded with tannic acid (TA), can be used to effectively treat myocardial ischemia and fibrosis in CMD mice, achieving an effect superior to that of free TA. The origin of this high therapeutic efficiency is revealed by transcriptomic and proteomic analyses. This investigation lays the groundwork for visual monitoring and the drug-targeted treatment of CMD.
Keywords: coronary microvascular dysfunction; ischemic myocardium; multimodal imaging; nanobubble; tannic acid.
© 2025 The Author(s). Advanced Healthcare Materials published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

