Treating Metabolic Dysregulation and Senescence by Caloric Restriction: Killing Two Birds with One Stone?
- PMID: 39857433
- PMCID: PMC11763027
- DOI: 10.3390/antiox14010099
Treating Metabolic Dysregulation and Senescence by Caloric Restriction: Killing Two Birds with One Stone?
Abstract
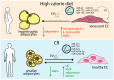
Cellular senescence is a state of permanent cell cycle arrest accompanied by metabolic activity and characteristic phenotypic changes. This process is crucial for developing age-related diseases, where excessive calorie intake accelerates metabolic dysfunction and aging. Overnutrition disturbs key metabolic pathways, including insulin/insulin-like growth factor signaling (IIS), the mammalian target of rapamycin (mTOR), and AMP-activated protein kinase. The dysregulation of these pathways contributes to insulin resistance, impaired autophagy, exacerbated oxidative stress, and mitochondrial dysfunction, further enhancing cellular senescence and systemic metabolic derangements. On the other hand, dysfunctional endothelial cells and adipocytes contribute to systemic inflammation, reduced nitric oxide production, and altered lipid metabolism. Numerous factors, including extracellular vesicles, mediate pathological communication between the vascular system and adipose tissue, amplifying metabolic imbalances. Meanwhile, caloric restriction (CR) emerges as a potent intervention to counteract overnutrition effects, improve mitochondrial function, reduce oxidative stress, and restore metabolic balance. CR modulates pathways such as IIS, mTOR, and sirtuins, enhancing glucose and lipid metabolism, reducing inflammation, and promoting autophagy. CR can extend the health span and mitigate age-related diseases by delaying cellular senescence and improving healthy endothelial-adipocyte interactions. This review highlights the crosstalk between endothelial cells and adipocytes, emphasizing CR potential in counteracting overnutrition-induced senescence and restoring vascular homeostasis.
Keywords: adipocytes; caloric restriction; endothelial dysfunction; oxidative stress; senescence.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Systemic aging fuels heart failure: Molecular mechanisms and therapeutic avenues.ESC Heart Fail. 2025 Apr;12(2):1059-1080. doi: 10.1002/ehf2.14947. Epub 2024 Jul 22. ESC Heart Fail. 2025. PMID: 39034866 Free PMC article. Review.
-
SGLT2 Inhibitors as Calorie Restriction Mimetics: Insights on Longevity Pathways and Age-Related Diseases.Endocrinology. 2021 Aug 1;162(8):bqab079. doi: 10.1210/endocr/bqab079. Endocrinology. 2021. PMID: 33857309
-
Caloric restriction mitigates age-associated senescence characteristics in subcutaneous adipose tissue-derived stem cells.Aging (Albany NY). 2024 May 9;16(9):7535-7552. doi: 10.18632/aging.205812. Epub 2024 May 9. Aging (Albany NY). 2024. PMID: 38728252 Free PMC article.
-
Calorie Restriction-Regulated Molecular Pathways and Its Impact on Various Age Groups: An Overview.DNA Cell Biol. 2022 May;41(5):459-468. doi: 10.1089/dna.2021.0922. Epub 2022 Apr 22. DNA Cell Biol. 2022. PMID: 35451872 Review.
-
Physiological Approaches Targeting Cellular and Mitochondrial Pathways Underlying Adipose Organ Senescence.Int J Mol Sci. 2023 Jul 19;24(14):11676. doi: 10.3390/ijms241411676. Int J Mol Sci. 2023. PMID: 37511435 Free PMC article. Review.
Cited by
-
The Hidden Price of Plenty: Oxidative Stress and Calorie-Induced Cardiometabolic Dysfunction.Life (Basel). 2025 Jun 27;15(7):1022. doi: 10.3390/life15071022. Life (Basel). 2025. PMID: 40724524 Free PMC article. Review.
-
Senescence of dental pulp stem cells: phenotypes, underlying mechanisms and regulatory molecules.Hum Cell. 2025 Jul 11;38(5):127. doi: 10.1007/s13577-025-01259-y. Hum Cell. 2025. PMID: 40646329 Review.
References
-
- Beyene H.B., Giles C., Huynh K., Wang T., Cinel M., Mellett N.A., Olshansky G., Meikle T.G., Watts G.F., Hung J., et al. Metabolic phenotyping of BMI to characterize cardiometabolic risk: Evidence from large population-based cohorts. Nat. Commun. 2023;14:6280. doi: 10.1038/s41467-023-41963-7. - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

