Evolution of Alzheimer's Disease Therapeutics: From Conventional Drugs to Medicinal Plants, Immunotherapy, Microbiotherapy and Nanotherapy
- PMID: 39861773
- PMCID: PMC11768419
- DOI: 10.3390/pharmaceutics17010128
Evolution of Alzheimer's Disease Therapeutics: From Conventional Drugs to Medicinal Plants, Immunotherapy, Microbiotherapy and Nanotherapy
Erratum in
-
Correction: Ortiz-Islas et al. Evolution of Alzheimer's Disease Therapeutics: From Conventional Drugs to Medicinal Plants, Immunotherapy, Microbiotherapy and Nanotherapy. Pharmaceutics 2025, 17, 128.Pharmaceutics. 2025 Dec 18;17(12):1625. doi: 10.3390/pharmaceutics17121625. Pharmaceutics. 2025. PMID: 41471143 Free PMC article.
Abstract
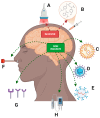
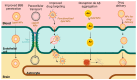
Alzheimer's disease (AD) represents an escalating global health crisis, constituting the leading cause of dementia among the elderly and profoundly impairing their quality of life. Current FDA-approved drugs, such as rivastigmine, donepezil, galantamine, and memantine, offer only modest symptomatic relief and are frequently associated with significant adverse effects. Faced with this challenge and in line with advances in the understanding of the pathophysiology of this neurodegenerative condition, various innovative therapeutic strategies have been explored. Here, we review novel approaches inspired by advanced knowledge of the underlying pathophysiological mechanisms of the disease. Among the therapeutic alternatives, immunotherapy stands out, employing monoclonal antibodies to specifically target and eliminate toxic proteins implicated in AD. Additionally, the use of medicinal plants is examined, as their synergistic effects among components may confer neuroprotective properties. The modulation of the gut microbiota is also addressed as a peripheral strategy that could influence neuroinflammatory and degenerative processes in the brain. Furthermore, the therapeutic potential of emerging approaches, such as the use of microRNAs to regulate key cellular processes and nanotherapy, which enables precise drug delivery to the central nervous system, is analyzed. Despite promising advances in these strategies, the incidence of Alzheimer's disease continues to rise. Therefore, it is proposed that achieving effective treatment in the future may require the integration of combined approaches, maximizing the synergistic effects of different therapeutic interventions.
Keywords: Alzheimer’s disease; fecal microbiota transplantation; immunotherapy; medicinal plants; miRNAs; nanotechnology; probiotics.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Yao D., Li T., Yu L., Hu M., He Y., Zhang R., Wu J., Li S., Kuang W., Yang X., et al. Selective degradation of hyperphosphorylated tau by proteolysis-targeting chimeras ameliorates cognitive function in Alzheimer’s disease model mice. Front. Pharmacol. 2024;15:1351792. doi: 10.3389/fphar.2024.1351792. - DOI - PMC - PubMed
-
- WHO. [(accessed on 12 December 2024)]. Available online: https://www.who.int/news-room/fact-sheets/detail/dementia.
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

