Prilling as an Effective Tool for Manufacturing Submicrometric and Nanometric PLGA Particles for Controlled Drug Delivery to Wounds: Stability and Curcumin Release
- PMID: 39861775
- PMCID: PMC11768656
- DOI: 10.3390/pharmaceutics17010129
Prilling as an Effective Tool for Manufacturing Submicrometric and Nanometric PLGA Particles for Controlled Drug Delivery to Wounds: Stability and Curcumin Release
Abstract
Background/objectives: This study investigates for the first time the use of the prilling technique in combination with solvent evaporation to produce nano- and submicrometric PLGA particles to deliver properly an active pharmaceutical ingredient. Curcumin (CCM), a hydrophobic compound classified under BCS (Biopharmaceutics Classification System) class IV, was selected as the model drug.
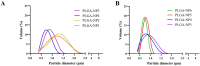
Methods: Key process parameters, including polymer concentration, solvent type, nozzle size, and surfactant levels, were optimized to obtain stable particles with a narrow size distribution determined by DLS analysis.
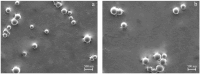
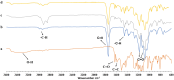
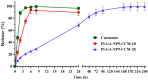
Results: Particles mean diameter (d50) 316 and 452 nm, depending on drug-loaded cargo as Curcumin-loaded PLGA nanoparticles demonstrated high encapsulation efficiency, assessed via HPLC analysis, stability, and controlled release profiles. In vitro studies revealed a faster release for lower drug loadings (90% release in 6 h) compared to sustained release over 7 days for higher-loaded nanoparticles, attributed to polymer degradation and drug-polymer interactions on the surface of the particles, as confirmed by FTIR analyses.
Conclusions: These findings underline the potential of this scalable technique for biomedical applications, offering a versatile platform for designing drug delivery systems with tailored release characteristics.
Keywords: PLGA; curcumin; nanoparticles; prilling; solvent evaporation; submicrometric particles.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

References
LinkOut - more resources
Full Text Sources

