Single-centre, double-blinded, randomised placebo-controlled trial to determine the effect of a 12-week home-based programme of footplate neuromuscular electrical stimulation on walking capacity in people with peripheral artery disease: a protocol for the Foot-PAD trial
- PMID: 39863411
- PMCID: PMC11784211
- DOI: 10.1136/bmjopen-2024-093162
Single-centre, double-blinded, randomised placebo-controlled trial to determine the effect of a 12-week home-based programme of footplate neuromuscular electrical stimulation on walking capacity in people with peripheral artery disease: a protocol for the Foot-PAD trial
Abstract
Introduction: Patients with peripheral artery disease (PAD) can experience intermittent claudication, which limits walking capacity and the ability to undertake daily activities. While exercise therapy is an established way to improve walking capacity in people with PAD, it is not feasible in all patients. Neuromuscular electrical stimulation (NMES) provides a way to passively induce repeated muscle contractions and has been widely used as a therapy for chronic conditions that limit functional capacity. Preliminary trials in patients with PAD demonstrate that stimulation of the leg muscles using a footplate-NMES device can be performed without pain and may lead to significant gains in walking capacity. Studies, to date, have been small and have not been adequately controlled to account for any potential placebo effect. Therefore, the current trial will compare the effect of a 12-week programme of footplate-NMES with a placebo-control on walking capacity (6 min walking distance) and other secondary outcomes in patients with PAD.
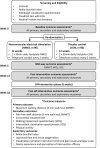
Methods and analysis: The Foot-PAD trial is a double-blinded, randomised placebo-controlled trial to determine the effect of a 12-week home-based programme of footplate NMES on walking capacity in people with PAD. This is a single-centre trial with numerous recruitment locations. A total of 180 participants with stable PAD and intermittent claudication will be randomly assigned (1:1 ratio) to receive either footplate-NMES (intervention condition) or footplate-placebo (control condition) for two 30 min periods each day for 12 weeks. The footplate-NMES device will deliver stimulation sufficient to induce contraction of the leg muscles and repeated plantar and dorsiflexion at the ankles. The footplate-placebo device will deliver a momentary low-intensity transient stimulation that is insufficient to induce contraction of the leg muscles. Outcomes will be assessed at baseline (week 0), mid-intervention (week 6), postintervention (week 12) and 6 weeks after the completion of the intervention (week 18). The primary outcome is walking capacity at week 12, measured as maximum walking distance during the 6 min walk test. Secondary outcomes will include pain-free walking distance during the 6 min walk test; pain-free and maximum walking time during a graded treadmill walking test; disease-specific quality of life (Intermittent Claudication Questionnaire), self-reported walking impairment (Walking Impairment Questionnaire) and accelerometer-derived physical activity levels. Exploratory outcomes will include the Ankle-Brachial Index; leg vascular function; perception of device-use experience and symptom monitoring throughout the trial using the Claudication Symptom Instrument and a pain Visual Analogue Scale.
Ethics and dissemination: The Foot-PAD trial has received ethics approval from the Human Research Ethics Committees of Queensland Health Metro North Hospital and Health Service (78962) and the University of the Sunshine Coast (A21659). Regardless of the study outcomes, the study findings will be published in peer-reviewed scientific journals and presented at scientific meetings.
Trial registration number: ACTRN12621001383853.
Keywords: Electric Stimulation Therapy; Exercise Test; Quality of Life; VASCULAR MEDICINE; VASCULAR SURGERY.
© Author(s) (or their employer(s)) 2025. Re-use permitted under CC BY-NC. No commercial re-use. See rights and permissions. Published by BMJ Group.
Conflict of interest statement
Competing interests: The authors declare no competing interests. The authors named on the protocol have no other relationship with Actegy (Funder), and no other financial or other competing interests that impact on their responsibilities towards the scientific value or potential publishing activities associated with the trial.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical