ESMO Clinical Practice Guideline interim update on the management of biliary tract cancer
- PMID: 39864891
- PMCID: PMC11846563
- DOI: 10.1016/j.esmoop.2024.104003
ESMO Clinical Practice Guideline interim update on the management of biliary tract cancer
Abstract
- •
This ESMO Clinical Practice Guideline update addresses new developments in the management of biliary tract cancer.
- •
Recommendations are given for first-line treatment with immune checkpoint inhibitors.
- •
Key recommendations are also provided for second-line treatment with targeted therapies.
- •
The update also covers the latest developments in molecular testing and intra-arterial therapies.
- •
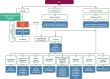
A management algorithm for early-stage, locally advanced and advanced/metastatic disease is provided.
Keywords: biliary tract cancer (BTC); guideline; molecular diagnostics; treatment.
Figures
References
-
- Vogel A., Bridgewater J., Edeline J., et al. Biliary tract cancer: ESMO Clinical Practice Guideline for diagnosis, treatment and follow-up. Ann Oncol. 2023;34(2):127–140. - PubMed
-
- Kendre G., Murugesan K., Brummer T., et al. Charting co-mutation patterns associated with actionable drivers in intrahepatic cholangiocarcinoma. J Hepatol. 2023;78(3):614–626. - PubMed
-
- Kelley R.K., Ueno M., Yoo C., et al. Pembrolizumab in combination with gemcitabine and cisplatin compared with gemcitabine and cisplatin alone for patients with advanced biliary tract cancer (KEYNOTE-966): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2023;401(10391):1853–1865. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

