Immunomodulation effects of collagen hydrogel encapsulating extracellular vesicles derived from calcium silicate stimulated-adipose mesenchymal stem cells for diabetic healing
- PMID: 39865263
- PMCID: PMC11770968
- DOI: 10.1186/s12951-025-03097-4
Immunomodulation effects of collagen hydrogel encapsulating extracellular vesicles derived from calcium silicate stimulated-adipose mesenchymal stem cells for diabetic healing
Abstract
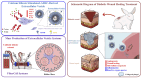
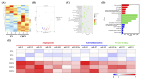
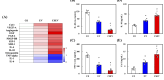
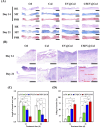
Diabetic wounds are characterized by chronic inflammation, reduced angiogenesis, and insufficient collagen deposition, leading to impaired healing. Extracellular vesicles (EVs) derived from adipose-derived mesenchymal stem cells (ADSC) offer a promising cell-free therapeutic strategy, yet their efficacy and immunomodulation can be enhanced through bioactivation. In this study, we developed calcium silicate (CS)-stimulated ADSC-derived EVs (CSEV) incorporated into collagen hydrogels to create a sustained-release system for promoting diabetic wound healing. CSEV exhibited enhanced protein content, surface marker expression, and bioactive cargo enriched with pro-angiogenic and anti-inflammatory factors. In vitro, CSEV-loaded collagen significantly reduced reactive oxygen species production, promoted cell proliferation and migration compared to standard EV-loaded collagen. Cytokine profiling revealed the upregulation of anti-inflammatory cytokines and extracellular matrix components, highlighting their immunomodulatory and regenerative potential. In vivo, histological evaluation of diabetic rabbit models treated with CSEV-loaded collagen revealed superior reepithelialization and organized collagen deposition, indicating accelerated wound closure. These findings underscore the potential of CSEV-loaded collagen hydrogels as an innovative and effective therapeutic platform for enhancing diabetic wound healing by simultaneously addressing inflammation and tissue regeneration.
Keywords: Calcium silicate; Diabetic wound healing; Extracellular vesicles; Immunomodulation.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: Not applicable. Consent for publication: Not applicable. Competing interests: Dr. Mei-Chih Chen is employed by Shine Out Bio Technology Co., Ltd., Taichung, Taiwan. All other authors confirm they have no conflicts of interest.
Figures





Similar articles
-
Lithium-doped calcium silicate scaffolds-activated M2-polarized macrophage-derived miR-145-5p-riched extracellular vesicles to enhance osteoimmunomodulation for accelerating bone regeneration.J Nanobiotechnology. 2025 Aug 25;23(1):586. doi: 10.1186/s12951-025-03679-2. J Nanobiotechnology. 2025. PMID: 40855489 Free PMC article.
-
Extracellular Vesicles Derived from Adipose-Derived Stem Cells Accelerate Diabetic Wound Healing by Suppressing the Expression of Matrix Metalloproteinase-9.Curr Pharm Biotechnol. 2022;23(6):894-901. doi: 10.2174/1389201022666210719154009. Curr Pharm Biotechnol. 2022. PMID: 34825636
-
Radial Egg White Hydrogel Releasing Extracellular Vesicles for Cell Fate Guidance and Accelerated Diabetic Skin Regeneration.Adv Healthc Mater. 2024 Dec;13(31):e2400016. doi: 10.1002/adhm.202400016. Epub 2024 Sep 17. Adv Healthc Mater. 2024. PMID: 39285803
-
Recent advances in the use of extracellular vesicles from adipose-derived stem cells for regenerative medical therapeutics.J Nanobiotechnology. 2024 Jun 6;22(1):316. doi: 10.1186/s12951-024-02603-4. J Nanobiotechnology. 2024. PMID: 38844939 Free PMC article. Review.
-
Extracellular vesicle-loaded hydrogels for tissue repair and regeneration.Mater Today Bio. 2022 Dec 21;18:100522. doi: 10.1016/j.mtbio.2022.100522. eCollection 2023 Feb. Mater Today Bio. 2022. PMID: 36593913 Free PMC article. Review.
Cited by
-
3D-printed magnesium/strontium-co-doped calcium silicate scaffolds promote angiogenesis and bone regeneration through synergistic bioactive ion stimulation.J Biol Eng. 2025 Jun 21;19(1):58. doi: 10.1186/s13036-025-00528-6. J Biol Eng. 2025. PMID: 40544274 Free PMC article.
-
Mechanical performance and aging resistance analysis of zinc oxide-reinforced polyurethane composites.RSC Adv. 2025 Aug 11;15(35):28358-28366. doi: 10.1039/d5ra03748d. eCollection 2025 Aug 11. RSC Adv. 2025. PMID: 40861968 Free PMC article.
-
Lithium-doped calcium silicate scaffolds-activated M2-polarized macrophage-derived miR-145-5p-riched extracellular vesicles to enhance osteoimmunomodulation for accelerating bone regeneration.J Nanobiotechnology. 2025 Aug 25;23(1):586. doi: 10.1186/s12951-025-03679-2. J Nanobiotechnology. 2025. PMID: 40855489 Free PMC article.
References
-
- Louiselle AE, Niemiec SM, Zgheib C, Liechty KW. Macrophage polarization and diabetic wound healing. Transl Res. 2021;236:109–16. - PubMed
-
- Zhang Y, Luo J, Zhang Q, Deng T. Growth factors, as biological macromolecules in bioactivity enhancing of electrospun wound dressings for diabetic wound healing: a review. Int J Biol Macromol. 2021;193:205–18. - PubMed
-
- Mirhaj M, Varshosaz J, Labbaf S, Emadi R, Seifalian AM, Sharifianjazi F, et al. Mupirocin loaded core-shell pluronic-pectin-keratin nanofibers improve human keratinocytes behavior, angiogenic activity and wound healing. Int J Biol Macromol. 2023;253:126700. - PubMed
MeSH terms
Substances
Grants and funding
- NSTC 113-2811-E-039-001/National Science and Technology Council
- NSTC 113-2314-B-039-018/National Science and Technology Council
- NSTC 113-2314-B-039-010/National Science and Technology Council
- NSTC 112-2628-E-039-001-MY3/National Science and Technology Council
- DMR-113-035/China Medical University Hospital
LinkOut - more resources
Full Text Sources

