Characterization of the Neurospora crassa GH72 family of Laminarin/Lichenin transferases and their roles in cell wall biogenesis
- PMID: 39866863
- PMCID: PMC11758075
- DOI: 10.1016/j.tcsw.2024.100140
Characterization of the Neurospora crassa GH72 family of Laminarin/Lichenin transferases and their roles in cell wall biogenesis
Abstract
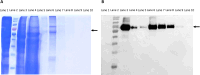
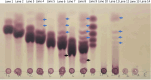
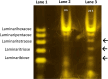
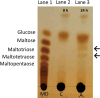
In Neurospora crassa vegetative hyphae, chitin, β-1,3-glucan (laminarin), and a mixed β-1,3-/β-1,4-glucan (lichenin) are the major cell wall polysaccharides. GH72 enzymes have been shown to function as β-1,3-glucanases and glucanosyltransferases and can function in crosslinking β-1,3-glucans together. To characterize the enzymatic activities of the N. crassa enzymes, we expressed GEL-1 with a HIS6 tag in N. crassa. A chimeric maltose binding protein:GEL-2 was produced in E. coli. Purified GEL-1 and GEL-2 were used to characterize their enzymatic activities. We employed thin-layer chromatography (TLC) and polyacrylamide carbohydrate gel electrophoresis (PACE) assays to visualize GEL-1 and GEL-2 hydrolase and transferase activities on lichenin and laminarin substrates. We determined that GEL-1 functions as a laminarinase (β-1,3-glucanase) and as a laminarin transferase. We found that GEL-2 can function as a laminarinase and as a licheninase (β-1,3-/β-1,4-mixed-glucanase) and can crosslink β-1,3-glucans together. We demonstrated that GEL-2 can form enzyme:lichenin intermediates, providing evidence that GEL-2 functions as a lichenin transferase as well as a β-1,3-glucan transferase and crosslinks both types of polysaccharides into the N. crassa cell wall.
Keywords: Fungal cell wall; GH72 family; Laminarin transferase; Lichenin transferase; Neurospora crassa.
© 2025 The Author(s).
Conflict of interest statement
The authors declare that they have no competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures






Similar articles
-
Characterization of Neurospora crassa GH16, GH17, and GH72 gene families of cell wall crosslinking enzymes.Cell Surf. 2022 Jan 15;8:100073. doi: 10.1016/j.tcsw.2022.100073. eCollection 2022 Dec. Cell Surf. 2022. PMID: 35079668 Free PMC article.
-
Neurospora crassa family GH72 glucanosyltransferases function to crosslink cell wall glycoprotein N-linked galactomannan to cell wall lichenin.Fungal Genet Biol. 2019 Feb;123:60-69. doi: 10.1016/j.fgb.2018.11.007. Epub 2018 Nov 29. Fungal Genet Biol. 2019. PMID: 30503329
-
Genetic and biochemical characterization of the GH72 family of cell wall transglycosylases in Neurospora crassa.Fungal Genet Biol. 2017 Apr;101:46-54. doi: 10.1016/j.fgb.2017.03.002. Epub 2017 Mar 8. Fungal Genet Biol. 2017. PMID: 28285007 Free PMC article.
-
The Genetics and Biochemistry of Cell Wall Structure and Synthesis in Neurospora crassa, a Model Filamentous Fungus.Front Microbiol. 2019 Oct 10;10:2294. doi: 10.3389/fmicb.2019.02294. eCollection 2019. Front Microbiol. 2019. PMID: 31649638 Free PMC article. Review.
-
X-ray crystal structures of Phanerochaete chrysosporium Laminarinase 16A in complex with products from lichenin and laminarin hydrolysis.FEBS J. 2009 Jul;276(14):3858-69. doi: 10.1111/j.1742-4658.2009.07099.x. Epub 2009 Jun 17. FEBS J. 2009. PMID: 19769746
References
-
- Blatzer M., Beauvais A., Henrissat B., Latge J.P. Revisiting old questions and new approaches to investigate the fungal cell wall construction. Curr. Top. Microbiol. Immunol. 2020;425:331–369. - PubMed
-
- Calderon J., Zavrel M., Ragni E., Fonzi W.A., Rupp S., Popolo L. PHR1, a pH-regulated gene of Candida albicans encoding a glucan-remodelling enzyme, is required for adhesion and invasion. Microbiology. 2010;156:2484–2494. - PubMed
LinkOut - more resources
Full Text Sources

