Elective Discontinuation of Larotrectinib in Pediatric Patients With TRK Fusion Sarcomas and Related Mesenchymal Tumors
- PMID: 39869835
- PMCID: PMC11949228
- DOI: 10.1200/JCO.24.00848
Elective Discontinuation of Larotrectinib in Pediatric Patients With TRK Fusion Sarcomas and Related Mesenchymal Tumors
Erratum in
-
Erratum: Elective Discontinuation of Larotrectinib in Pediatric Patients With TRK Fusion Sarcomas and Related Mesenchymal Tumors.J Clin Oncol. 2025 Mar 10;43(8):1046. doi: 10.1200/JCO-25-00252. Epub 2025 Feb 11. J Clin Oncol. 2025. PMID: 39951654 Free PMC article. No abstract available.
-
Erratum: Elective Discontinuation of Larotrectinib in Pediatric Patients With TRK Fusion Sarcomas and Related Mesenchymal Tumors.J Clin Oncol. 2025 Apr;43(10):1270. doi: 10.1200/JCO-25-00353. Epub 2025 Mar 3. J Clin Oncol. 2025. PMID: 40030088 Free PMC article. No abstract available.
Abstract
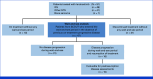
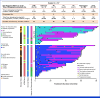
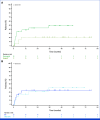
Larotrectinib is a highly selective tropomyosin receptor kinase (TRK) inhibitor with efficacy in children with TRK fusion tumors. We evaluated patient outcomes after elective discontinuation of larotrectinib in the absence of disease progression in a protocol-defined wait-and-see subset analysis of eligible patients where treatment resumption with larotrectinib was allowed if disease progressed. We also assessed the safety and efficacy of larotrectinib in all pediatric patients with sarcoma. This cohort included 91 patients (younger than 18 years) from two clinical trials: infantile fibrosarcoma (49), other soft tissue sarcomas or related mesenchymal tumors (41), and bone sarcoma (1). Treatment-related adverse events were of maximum grade 1 or 2 in 25% and 25% of patients, respectively. The overall response rate was 87% (95% CI, 78 to 93). In the wait-and-see analysis, 47 patients discontinued larotrectinib. Median time from discontinuation to disease progression was not reached. Sixteen patients had tumor progression during the wait-and-see period. All 16 patients resumed larotrectinib, and 15 (94%) achieved disease control, with 11 objective responses. Larotrectinib continues to demonstrate durable responses with favorable safety in children with TRK fusion sarcomas. Treatment discontinuation is feasible in select patients with objective response and clinical benefit noted in those who have disease progression after elective treatment discontinuation.
Trial registration: ClinicalTrials.gov NCT02576431 NCT02637687.
Conflict of interest statement
The following represents disclosure information provided by authors of this manuscript. All relationships are considered compensated unless otherwise noted. Relationships are self-held unless noted. I = Immediate Family Member, Inst = My Institution. Relationships may not relate to the subject matter of this manuscript. For more information about ASCO's conflict of interest policy, please refer to
Open Payments is a public database containing information reported by companies about payments made to US-licensed physicians (
No other potential conflicts of interest were reported.
Figures
References
-
- Davis JL, Lockwood CM, Stohr B, et al. : Expanding the spectrum of pediatric NTRK-rearranged mesenchymal tumors. Am J Surg Pathol 43:435-445, 2018 - PubMed
-
- Lemelle L, Guillemot D, Hermann AL, et al. : Neurotrophic tropomyosin receptor kinase (NTRK) fusion positive tumors: A historical cohort analysis. Expert Rev Anticancer Ther 23:865-874, 2023 - PubMed

