Early retinoic acid signaling organizes the body axis and defines domains for the forelimb and eye
- PMID: 39870430
- PMCID: PMC11969570
- DOI: 10.1016/bs.ctdb.2024.10.002
Early retinoic acid signaling organizes the body axis and defines domains for the forelimb and eye
Abstract
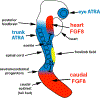
All-trans RA (ATRA) is a small molecule derived from retinol (vitamin A) that directly controls gene expression at the transcriptional level by serving as a ligand for nuclear ATRA receptors. ATRA is produced by ATRA-generating enzymes that convert retinol to retinaldehyde (retinol dehydrogenase; RDH10) followed by conversion of retinaldehyde to ATRA (retinaldehyde dehydrogenase; ALDH1A1, ALDH1A2, or ALDH1A3). Determining what ATRA normally does during vertebrate development has been challenging as studies employing ATRA gain-of-function (RA treatment) often do not agree with genetic loss-of-function studies that remove ATRA via knockouts of ATRA-generating enzymes. In mouse embryos, ATRA is first generated at stage E7.5 by ATRA-generating enzymes whose genes are first expressed at that stage. This article focuses upon what ATRA normally does at early stages based upon these knockout studies. It has been observed that early-generated ATRA performs three essential functions: (1) activation of genes that control hindbrain and spinal cord patterning; (2) repression of Fgf8 in the heart field and caudal progenitors to provide an FGF8-free region in the trunk essential for somitogenesis, heart morphogenesis, and initiation of forelimb fields; and (3) actions that stimulate invagination of the optic vesicle to form the optic cup.
Keywords: Body axis formation; Eye morphogenesis; Forelimb initiation; Neural patterning; Retinoic acid.
Copyright © 2025. Published by Elsevier Inc.
Figures

Similar articles
-
Towards a Better Vision of Retinoic Acid Signaling during Eye Development.Cells. 2022 Jan 19;11(3):322. doi: 10.3390/cells11030322. Cells. 2022. PMID: 35159132 Free PMC article. Review.
-
Investigation of retinoic acid function during embryonic brain development using retinaldehyde-rescued Rdh10 knockout mice.Dev Dyn. 2013 Sep;242(9):1056-65. doi: 10.1002/dvdy.23999. Epub 2013 Jul 22. Dev Dyn. 2013. PMID: 23765990 Free PMC article.
-
Dhrs3 protein attenuates retinoic acid signaling and is required for early embryonic patterning.J Biol Chem. 2013 Nov 1;288(44):31477-87. doi: 10.1074/jbc.M113.514984. Epub 2013 Sep 17. J Biol Chem. 2013. PMID: 24045938 Free PMC article.
-
Keeping an eye on retinoic acid signaling during eye development.Chem Biol Interact. 2009 Mar 16;178(1-3):178-81. doi: 10.1016/j.cbi.2008.09.004. Epub 2008 Sep 11. Chem Biol Interact. 2009. PMID: 18831967 Free PMC article.
-
Retinoic acid regulation of the somitogenesis clock.Birth Defects Res C Embryo Today. 2007 Jun;81(2):84-92. doi: 10.1002/bdrc.20092. Birth Defects Res C Embryo Today. 2007. PMID: 17600781 Free PMC article. Review.
References
-
- Agarwal P, Wylie JN, Galceran J, Arkhitko O, Li C, Deng C, et al. (2003). Tbx5 is essential for forelimb bud initiation following patterning of the limb field in the mouse embryo. Development, 130, 623–633. - PubMed
-
- Ahn Y, Mullan HE, & Krumlauf R (2014). Long-range regulation by shared retinoic acid response elements modulates dynamic expression of posterior Hoxb genes in CNS development. Developmental Biology, 388, 134–144. - PubMed
-
- Begemann G, Schilling TF, Rauch GJ, Geisler R, & Ingham PW (2001). The zebrafish neckless mutation reveals a requirement for raldh2 in mesodermal signals that pattern the hindbrain. Development, 128, 3081–3094. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

