The transcriptional response of cortical neurons to concussion reveals divergent fates after injury
- PMID: 39870620
- PMCID: PMC11772587
- DOI: 10.1038/s41467-025-56292-0
The transcriptional response of cortical neurons to concussion reveals divergent fates after injury
Abstract
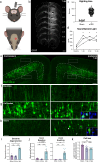
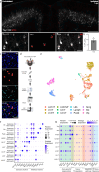
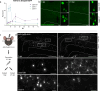
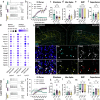
Traumatic brain injury (TBI) is a risk factor for neurodegeneration, however little is known about how this kind of injury alters neuron subtypes. In this study, we follow neuronal populations over time after a single mild TBI (mTBI) to assess long ranging consequences of injury at the level of single, transcriptionally defined neuronal classes. We find that the stress-responsive Activating Transcription Factor 3 (ATF3) defines a population of cortical neurons after mTBI. Using an inducible reporter linked to ATF3, we genetically mark these damaged cells to track them over time. We find that a population in layer V undergoes cell death acutely after injury, while another in layer II/III survives long term and remains electrically active. To investigate the mechanism controlling layer V neuron death, we genetically silenced candidate stress response pathways. We found that the axon injury responsive dual leucine zipper kinase (DLK) is required for the layer V neuron death. This work provides a rationale for targeting the DLK signaling pathway as a therapeutic intervention for traumatic brain injury. Beyond this, our approach to track neurons after a mild, subclinical injury can inform our understanding of neuronal susceptibility to repeated impacts.
© 2025. This is a U.S. Government work and not under copyright protection in the US; foreign copyright protection may apply.
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures

Update of
-
The transcriptional response of cortical neurons to concussion reveals divergent fates after injury.bioRxiv [Preprint]. 2024 Sep 17:2024.02.26.581939. doi: 10.1101/2024.02.26.581939. bioRxiv. 2024. Update in: Nat Commun. 2025 Jan 27;16(1):1097. doi: 10.1038/s41467-025-56292-0. PMID: 38463961 Free PMC article. Updated. Preprint.
References
-
- Maas, A. I. R. et al. Traumatic brain injury: integrated approaches to improve prevention, clinical care, and research. Lancet Neurol.16, 987–1048 (2017). - PubMed
MeSH terms
Substances
Grants and funding
- ZIA HD008966/ImNIH/Intramural NIH HHS/United States
- ZIA MH002970/ImNIH/Intramural NIH HHS/United States
- ZIC HD008986/ImNIH/Intramural NIH HHS/United States
- 1ZICHD008986-05/U.S. Department of Health & Human Services | NIH | Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD)
- ZIA-HD008966/U.S. Department of Health & Human Services | NIH | Eunice Kennedy Shriver National Institute of Child Health and Human Development (NICHD)
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Miscellaneous

