Programmable scanning diffuse speckle contrast imaging of cerebral blood flow
- PMID: 39872020
- PMCID: PMC11770344
- DOI: 10.1117/1.NPh.12.1.015006
Programmable scanning diffuse speckle contrast imaging of cerebral blood flow
Abstract
Significance: Cerebral blood flow (CBF) imaging is crucial for diagnosing cerebrovascular diseases. However, existing large neuroimaging techniques with high cost, low sampling rate, and poor mobility make them unsuitable for continuous and longitudinal CBF monitoring at the bedside.
Aim: We aimed to develop a low-cost, portable, programmable scanning diffuse speckle contrast imaging (PS-DSCI) technology for fast, high-density, and depth-sensitive imaging of CBF in rodents.
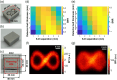
Approach: The PS-DSCI employed a programmable digital micromirror device (DMD) for remote line-shaped laser (785 nm) scanning on tissue surface and synchronized a 2D camera for capturing boundary diffuse laser speckle contrasts. New algorithms were developed to address deformations of line-shaped scanning, thus minimizing CBF reconstruction artifacts. The PS-DSCI was examined in head-simulating phantoms and adult mice.
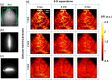
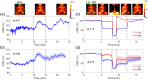
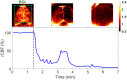
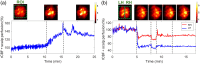
Results: The PS-DSCI enables resolving intralipid particle flow contrasts at different tissue depths. In vivo experiments in adult mice demonstrated the capability of PS-DSCI to image global/regional CBF variations induced by 8% inhalation and transient carotid artery ligations.
Conclusions: Compared with conventional point scanning, line scanning in PS-DSCI significantly increases spatiotemporal resolution. The high sampling rate of PS-DSCI is crucial for capturing rapid CBF changes while high spatial resolution is important for visualizing brain vasculature.
Keywords: cerebral blood flow; diffuse optics; digital micromirror device; line-shaped scanning; speckle contrast imaging.
© 2025 The Authors.
Figures



Update of
-
Programmable scanning diffuse speckle contrast imaging of cerebral blood flow.ArXiv [Preprint]. 2024 Aug 22:arXiv:2408.12715v1. ArXiv. 2024. Update in: Neurophotonics. 2025 Jan;12(1):015006. doi: 10.1117/1.NPh.12.1.015006. PMID: 39253639 Free PMC article. Updated. Preprint.
References
Grants and funding
LinkOut - more resources
Full Text Sources

