Comparative transcriptomic rhythms in the mouse and human prefrontal cortex
- PMID: 39872996
- PMCID: PMC11769989
- DOI: 10.3389/fnins.2024.1524615
Comparative transcriptomic rhythms in the mouse and human prefrontal cortex
Abstract
Introduction: Alterations in multiple subregions of the human prefrontal cortex (PFC) have been heavily implicated in psychiatric diseases. Moreover, emerging evidence suggests that circadian rhythms in gene expression are present across the brain, including in the PFC, and that these rhythms are altered in disease. However, investigation into the potential circadian mechanisms underlying these diseases in animal models must contend with the fact that the human PFC is highly evolved and specialized relative to that of rodents.
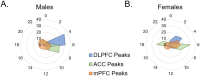
Methods: Here, we use RNA sequencing to lay the groundwork for translational studies of molecular rhythms through a sex-specific, cross species comparison of transcriptomic rhythms between the mouse medial PFC (mPFC) and two subregions of the human PFC, the anterior cingulate cortex (ACC) and the dorsolateral PFC (DLPFC).
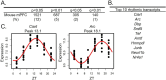
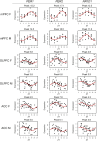
Results: We find that while circadian rhythm signaling is conserved across species and subregions, there is a phase shift in the expression of core clock genes between the mouse mPFC and human PFC subregions that differs by sex. Furthermore, we find that the identity of rhythmic transcripts is largely unique between the mouse mPFC and human PFC subregions, with the most overlap (20%, 236 transcripts) between the mouse mPFC and the human ACC in females. Nevertheless, we find that basic biological processes are enriched for rhythmic transcripts across species, with key differences between regions and sexes.
Discussion: Together, this work highlights both the evolutionary conservation of transcriptomic rhythms and the advancement of the human PFC, underscoring the importance of considering cross-species differences when using animal models.
Keywords: circadian rhythms; human post mortem tissue; mouse; prefrontal cortex; transcriptomics.
Copyright © 2025 Burns, Jenkins, Xue, Petersen, Ketchesin, Perez, Vadnie, Scott, Seney, Tseng and McClung.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures

Update of
-
Comparative transcriptomic rhythms in the mouse and human prefrontal cortex.bioRxiv [Preprint]. 2024 Sep 26:2024.09.26.615154. doi: 10.1101/2024.09.26.615154. bioRxiv. 2024. Update in: Front Neurosci. 2025 Jan 13;18:1524615. doi: 10.3389/fnins.2024.1524615. PMID: 39386590 Free PMC article. Updated. Preprint.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
Miscellaneous

