Advancing osseointegration research: A dynamic three-dimensional (3D) in vitro culture model for dental implants
- PMID: 39873044
- PMCID: PMC11763192
- DOI: 10.1016/j.jds.2024.06.018
Advancing osseointegration research: A dynamic three-dimensional (3D) in vitro culture model for dental implants
Abstract
Background/purpose: In-vitro studies are essential for understanding cellular responses, but traditional culture systems often neglect the three-dimensional (3D) structure of real implants, leading to limitations in cellular recruitment and behavior largely governed by gravity. The objective of this study was to pioneer a novel 3D dynamic osteoblastic culture system for assessing the biological capabilities of dental implants in a more clinically and physiologically relevant manner.
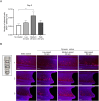
Materials and methods: Rat bone marrow-derived osteoblasts were cultured in a 24-well dish with a vertically positioned dental implant. Controlled rotation using a 3D rotator with 3° tilts was applied. Cell attachment, proliferation, and differentiation on implant surfaces were evaluated in response to different surface topographies, physicochemical properties, and local environments.
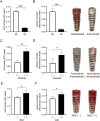
Results: Among the tested rotational speeds (0, 10, 30, 50 rpm), optimal osteoblast attachment and proliferation were observed at 30 rpm. A linear correlation was found between cell attachment and rotation speed up to 30 rpm, declining at 50 rpm. Alkaline phosphatase (ALP) activity and mineralized matrix formation were elevated on newly acid-etched, hydrophilic surfaces compared to their 4-week-old hydrophobic surfaces. Sandblasted implants showed higher ALP activity and matrix mineralization. Adding N-acetyl cysteine to the culture medium increased ALP activity and mineralization.
Conclusion: Osteoblasts successfully attached, proliferated, and mineralized on dental implants in vitro under optimized dynamic conditions. This system differentiated the biological capabilities of implants with varying surface topographies, wettability, and biochemically modulated environments. These findings support developing a 3D dynamic dental implant culture model, advancing osseointegration research and innovating dental implant designs.
Keywords: Hydrophilic materials; Osseointegration; Osteoblasts; Surface properties.
© 2024 Association for Dental Sciences of the Republic of China. Publishing services by Elsevier B.Vé.
Conflict of interest statement
The authors have no conflicts of interest relevant to this article.
Figures





References
-
- Schneider G.B., Perinpanayagam H., Clegg M., et al. Implant surface roughness affects osteoblast gene expression. J Dent Res. 2003;82:372–376. - PubMed
-
- Cooper L.F., Masuda T., Yliheikkila P.K., Felton D.A. Generalizations regarding the process and phenomenon of osseointegration. Part II. In vitro studies. Int J Oral Maxillofac Implants. 1998;13:163–174. - PubMed
-
- Wennerberg A., Albrektsson T. Suggested guidelines for the topographic evaluation of implant surfaces. Int J Oral Maxillofac Implants. 2000;15:331–344. - PubMed
-
- Wennerberg A., Albrektsson T. Effects of titanium surface topography on bone integration: a systematic review. Clin Oral Implants Res. 2009;20(Suppl 4):172–184. - PubMed
-
- Masaki C., Schneider G.B., Zaharias R., Seabold D., Stanford C. Effects of implant surface microtopography on osteoblast gene expression. Clin Oral Implants Res. 2005;16:650–656. - PubMed

