Robotic Versus Open Placement of Hepatic Artery Infusion Pumps
- PMID: 39875717
- PMCID: PMC11976338
- DOI: 10.1245/s10434-025-16915-8
Robotic Versus Open Placement of Hepatic Artery Infusion Pumps
Abstract
Background: A growing number of centers offer hepatic artery infusion pump (HAIP) chemotherapy for advanced liver malignancies. While small series have demonstrated feasibility of robotic HAIP placement, comparison of outcomes with open placement is lacking. We compared outcomes after robotic versus open HAIP placement.
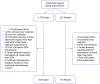
Methods: We retrospectively reviewed HAIP placement without concurrent hepatectomy at Memorial Sloan Kettering Cancer Center from 1 January 2011 to 15 September 2022, and Duke Health from 1 November 2018 to 18 May 2023. Patients with prior liver surgery or who required catheterization of a non-standard vessel were excluded. Propensity score matching weights (PSMW) were calculated using age, sex, race, body mass index, American Society of Anesthesiologists class, neoadjuvant chemotherapy, colorectal procedure, and institution. Survey-weighted generalized linear models assessed the relationship between approach and outcomes.
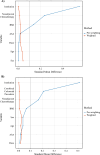
Results: Of 2002 consecutive HAIP placements, 819 (645 open/174 robotic) met the inclusion criteria. A higher proportion of open procedures involved combined colorectal procedures; other patient characteristics were similar. Overall, 15% of patients experienced an HAIP-specific complication and 12% required re-intervention; 2.7% had HAIP failure ≤ 90 days. After PSMW, the robotic approach had a longer operative time (β = 68 min, 95% confidence interval [CI] 55-81, p < 0.001) but shorter length of stay (β = - 1.8 days, 95% CI - 2.3 to 1.3, p < 0.001). The robotic approach was associated with increased HAIP-specific complications (odds ratio [OR] 1.72, p = 0.025) and re-intervention (OR 2.33, p < 0.001), with no difference in time to initiation of HAIP chemotherapy or HAIP failure.
Conclusions: Robotic HAIP placement was associated with increased postoperative complications and significantly shorter length of stay, with similar time to initiation of HAIP therapy. There was no difference in the rate of early HAIP failure versus the open approach. These results suggest robotic HAIP placement is feasible and effective.
Keywords: Colorectal cancer liver metastasis; HAIP; Hepatic artery infusion pump; Intrahepatic cholangiocarcinoma; Liver cancer; Liver-directed therapy; Propensity score matching weights; Robotic HAIP placement; Robotic surgery.
© 2025. The Author(s).
Conflict of interest statement
Disclosures: Alice C. Wei reports receiving consulting fees from Histosonics and institutional clinical trial funding from Ipsen. Sabino Zani reports consulting for Asensus Surgical. Jeffrey Drebin reports equity shares in ALNY, IONS and ARWR, purchased from a broker. Lauren E. Schleimer, Annie Liu, Hannah L. Kalvin, Ahmad Bashir Barekzai, Ankur P. Choubey, Joslyn Jung, Rubiya Haque, William R. Jarnagin, Vinod P. Balachandran, Ruben Geevarghese, Brett Marinelli, Mithat Gonen, Peter J. Allen, Michael I. D’Angelica, T. Peter Kingham, Michael E. Lidsky, and Kevin C. Soares have no conflicts of interest disclosures to report that may be relevant to the contents of this study.
Figures

References
-
- Franssen S, Rousian M, Nooijen LE, et al. Hepatic arterial infusion pump chemotherapy in patients with advanced intrahepatic cholangiocarcinoma confined to the liver: a multicenter phase II trial. J Clin Oncol. 2024;42(3 Suppl):433.
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

