Enhanced Antitumor Efficacy and Reduced Toxicity in Colorectal Cancer Using a Novel Multifunctional Rg3- Targeting Nanosystem Encapsulated with Oxaliplatin and Calcium Peroxide
- PMID: 39877588
- PMCID: PMC11774109
- DOI: 10.2147/IJN.S502076
Enhanced Antitumor Efficacy and Reduced Toxicity in Colorectal Cancer Using a Novel Multifunctional Rg3- Targeting Nanosystem Encapsulated with Oxaliplatin and Calcium Peroxide
Abstract
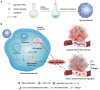
Purpose: Colorectal cancer (CRC) is the second leading cause of cancer-related deaths worldwide. Oxaliplatin (OXA) is currently the primary chemotherapeutic agent for CRC, but its efficacy is limited by the tumor microenvironment (TME). Here, we present a combined approach of chemotherapy and TME modulation for CRC treatment. A multifunctional nanosystem (Rg3-Lip-OXA/CaO2) was established using Ginsenoside Rg3 liposomes targeting glucose transporter 1 overexpressed on the surface of CRC cells to co-deliver OXA and calcium peroxide (CaO2).
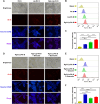
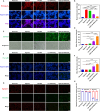
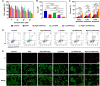
Methods: The CaO2 nanoparticles were synthesized via the CaCl2-H2O2 reaction under alkaline conditions and characterized using X-ray diffraction (XRD) and X-ray photoelectron spectroscopy (XPS). Rg3-Lip-OXA/CaO2 was prepared through a thin-film hydration approach and characterized; additionally, its stability and release behavior were studied. The O2, H2O2, and Ca2+ generation ability of Rg3-Lip-OXA/CaO2 in solution and HCT116 cells were measured. The in vitro cellular uptake was observed via fluorescence microscope and flow cytometry. In vitro cytotoxicity was evaluated using the CCK-8 assay, flow cytometry, and live/dead cell staining. The in vivo targeting effect as well as antitumor efficacy were determined in HCT116 tumor-bearing mice. Finally, the acute toxicity of Rg3-Lip-OXA/CaO2 was investigated in ICR mice to explore its safety.
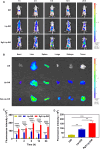
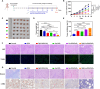
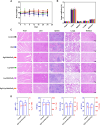
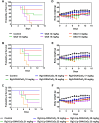
Results: The XRD and XPS analyses confirmed the successful synthesis of CaO2 nanoparticles. The Rg3-Lip-OXA/CaO2 exhibited an average particle size of approximately 92.98 nm with good stability and sustained release behavior. In vitro and in vivo studies confirmed optimal targeting by Rg3-Lip and demonstrated that the nanosystem effectively produced O2, H2O2 and Ca2+, resulting in significant cytotoxicity. Additionally, in vivo studies revealed substantial tumor growth suppression and reduced tumor-associated fibroblasts (TAFs) and collagen. Acute toxicity studies indicated that Rg3-Lip-OXA/CaO2 markedly reduced the toxicity of chemotherapeutic drugs.
Conclusion: This multifunctional nanosystem enhances chemotherapy efficacy and reduces toxicity, offering a promising approach for optimizing CRC treatment and potential clinical application.
Keywords: anticancer therapy; calcium peroxide; multifunctional nanosystem; oxaliplatin; targeted drug delivery; tumor microenvironment.
© 2025 Xie et al.
Conflict of interest statement
The authors report no conflicts of interest in this work.
Figures

References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
Medical
Miscellaneous

