Scratching promotes allergic inflammation and host defense via neurogenic mast cell activation
- PMID: 39883751
- PMCID: PMC11983162
- DOI: 10.1126/science.adn9390
Scratching promotes allergic inflammation and host defense via neurogenic mast cell activation
Abstract
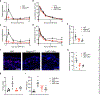
Itch is a dominant symptom in dermatitis, and scratching promotes cutaneous inflammation, thereby worsening disease. However, the mechanisms through which scratching exacerbates inflammation and whether scratching provides benefit to the host are largely unknown. We found that scratching was required for skin inflammation in mouse models dependent on FcεRI-mediated mast cell activation. Scratching-induced inflammation required pain-sensing nociceptors, the neuropeptide substance P, and the mast cell receptor MrgprB2. Scratching also increased cutaneous inflammation and augmented host defense to superficial Staphylococcus aureus infection. Thus, through the activation of nociceptor-driven neuroinflammation, scratching both exacerbated allergic skin disease and provided protection from S. aureus, reconciling the seemingly paradoxical role of scratching as a pathological process and evolutionary adaptation.
Conflict of interest statement
Figures
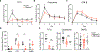



Comment in
-
Scratching more than an itch.Science. 2025 Jan 31;387(6733):473-474. doi: 10.1126/science.adv1573. Epub 2025 Jan 30. Science. 2025. PMID: 39883786
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

