Automated insulin delivery during the first 6 months postpartum (AiDAPT): a prespecified extension study
- PMID: 39884300
- PMCID: PMC7617783
- DOI: 10.1016/S2213-8587(24)00340-1
Automated insulin delivery during the first 6 months postpartum (AiDAPT): a prespecified extension study
Abstract
Background: Clinical guidelines in the UK and elsewhere do not specifically address hybrid closed loop (HCL) use in the postpartum period when the demands of caring for a newborn are paramount. Our aim was to evaluate the safety and efficacy of HCL use during the first 6 months postpartum compared with standard care.
Methods: In this prespecified extension to a multicentre, randomised controlled trial, pregnant women with type 1 diabetes at nine UK sites were followed up for 6 months postpartum. Eligible participants (AiDAPT participants recruited after the implementation of the postpartum protocol amendment approval, those still pregnant or within six months of delivery at the time of amendment implementation and still using HCL or continuous glucose monitoring [CGM] therapy) continued their randomly assigned treatment, either standard insulin therapy with CGM or HCL therapy (CamAPS FX system version 0.3.1, CamDiab, Cambridge, UK). Participants were randomised in a 1:1 ratio with stratification by clinical site using randomly permuted block sizes of 2 or 4. The primary outcome was the between-group difference in percentage time in range ([TIR] 3·9-10·0 mmol/L [70-180mg/dL]), measured during the periods of month 0 up to 3, months 3 to 6, and over 6 months postpartum. The study is registered at ClinicalTrials.gov (ISRCTN56898625) and is complete.
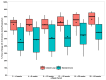
Findings: Of the 124 AiDAPT trial participants, 66 (53%) were ineligible for inclusion in the postpartum extension, and 57 participants consented to continue their treatment per original random allocation. The mean age was 31 years (SD 4), and all participants had early pregnancy HbA1c 59·4 mmol/mol (SD 10·5 [7·6% SD 1·0%]). In the 6 months postpartum, mean time with glucose levels within the target range was higher in the HCL group compared with the standard care group (72% [SD 12%] vs 54% [17%]), with an adjusted treatment difference of 15% (95% CI 7 to 22). Results for hyperglycaemia (>10·0 mmol/L) and mean CGM glucose also favoured HCL (-14% [95% CI -23% to -6%] and -1·3 mmol/L [-2·3 to -0·3], respectively). Hypoglycaemia rates were low, with no between-group differences (2·4% vs 2·6%). There were no treatment effect changes depending on postpartum period (0 up to 3 months vs 3 to 6 months) and no unanticipated safety problems.
Interpretation: Participants in the HCL group maintained 70% TIR during the first 6 months postpartum, supporting continued use of HCL rather than standard insulin therapy for people with diabetes once they have given birth.
Funding: National Institute for Health Research, Juvenile Diabetes Research Foundation, and Diabetes Research & Wellness Foundation. CGM devices were provided by Dexcom at a discounted price.
Copyright © 2025 The Author(s). Published by Elsevier Ltd. This is an Open Access article under the CC BY 4.0 license. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of interests HRM sits on the Medtronic European Scientific Advisory Board and reports speaker honoraria from Dexcom, Abbott, Medtronic, Novo Nordisk, and Ypsomed. EMS reports receiving speaker honoraria from Abbott Diabetes Care and Eli Lilly. RH reports receiving speaker honoraria from Eli Lilly, Dexcom, and Novo Nordisk, receiving license or consultancy fees from B Braun and Abbott Diabetes Care; patents related to closed‑loop systems, and being a director at CamDiab. MEW reports patents related to closed‑loop systems, and being a consultant at CamDiab. SH is a UK member of the Medtronic Advisory Board, reports being a consultant at CamDiab, and providing training for Dexcom. Dexcom was supplied continuous glucose monitoring (CGM) systems at reduced cost.
Figures
Comment in
-
Automated insulin delivery postpartum: insights from the AiDAPT study extension.Lancet Diabetes Endocrinol. 2025 Mar;13(3):171-172. doi: 10.1016/S2213-8587(24)00374-7. Epub 2025 Jan 27. Lancet Diabetes Endocrinol. 2025. PMID: 39884301 No abstract available.
References
-
- Lev-Ran A. Sharp temporary drop in insulin requirement after cesarean section in diabetic patients. Am J Obstet Gynecol. 1974;120:905–8. - PubMed
-
- Achong N, Duncan EL, McIntyre HD, Callaway L. Peripartum management of glycemia in women with type 1 diabetes. Diabetes Care. 2014;37:364–71. - PubMed
-
- Ringholm L, Mathiesen ER, Kelstrup L, Damm P. Managing type 1 diabetes mellitus in pregnancy--from planning to breastfeeding. Nat Rev Endocrinol. 2012;8:659–67. - PubMed
-
- Dashora U, Murphy HR, Temple RC, et al. Managing hyperglycaemia during antenatal steroid administration, labour and birth in pregnant women with diabetes. Diabet Med J Br Diabet Assoc. 2018;35:1005–10. - PubMed
-
- Nørgaard SK, Nørgaard K, Roskjær AB, Mathiesen ER, Ringholm L. Insulin Pump Settings During Breastfeeding in Women with Type 1 Diabetes. Diabetes Technol Ther. 2020;22:314–20. - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

