Biomimetic polydopamine loaded with janus kinase inhibitor for synergistic vitiligo therapy via hydrogel microneedles
- PMID: 39885576
- PMCID: PMC11780829
- DOI: 10.1186/s12951-025-03119-1
Biomimetic polydopamine loaded with janus kinase inhibitor for synergistic vitiligo therapy via hydrogel microneedles
Abstract
Background: Both oxidative stress and autoimmune responses play crucial roles in the development of vitiligo. Under oxidative stress, the apoptotic melanocytes expose self-antigens and release high mobility group box 1 (HMGB1), triggering autoimmune activation and recruiting CD8+ T cells. This process further leads to the destruction of melanocytes, resulting in the lack of melanin granules. Additionally, the accumulated CD8+ T cells release interferon-γ (IFN-γ) to activate janus kinase-signal transducer and activator of transcription (JAK-STAT) pathway in keratinocytes. Both oxidative stress and IFN-γ-JAK-STAT activation induce keratinocytes to express and release T cell chemotactic factors, exacerbating the process of vitiligo. Reducing the accumulation of CD8+ T cells by safeguarding melanocytes and keratinocytes from oxidative stress may be contemplated as a promising approach for vitiligo therapy.
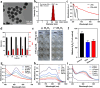
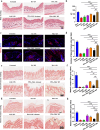
Results: In this study, we introduce a novel therapeutic agent called PDA-JAKi, which is capable of both eliminating oxidative stress and inhibiting T cell activation. Specifically, we have incorporated the janus kinase inhibitor (JAKi) tofacitinib into antioxidant polydopamine (PDA) nanoparticles, resulting in the formation of uniform PDA-JAKi nanodrug. PDA-JAKi effectively mitigates oxidative stress-induced apoptosis in melanocytes, reducing the antigen presentation and release of HMGB1. In addition, PDA-JAKi simultaneously attenuates oxidative stress and blocks the IFN-γ-JAK-STAT pathway to reduce the expression of C-X-C motif chemokine ligand 9/10/16 (CXCL9/10/16) in keratinocytes. We precisely deliver this therapeutic agent to the dermis using microneedle (MN) patches, aiming to enhance therapeutic efficacy compared to traditional drug administration methods. After PDA-JAKi MN treatment, the symptoms of vitiligo in mice are alleviated, and the affected areas regain pigmentation. Enhancements have been observed in the dermal thickness, the numbers of melanocytes and the content of melanin within the treated skin area. Moreover, there is a notable reduction in reactive oxygen species (ROS) level. Concurrently, substantial decreases were noted in CD8+ T cell infiltration, as well as the levels of IFN-γ and chemotactic factors CXCL9/10/16.
Conclusions: In summary, PDA-JAKi MN patches emerge as a promising therapeutic agent for vitiligo treatment.
Keywords: JAK inhibitor; Microneedles; Oxidative stress; Polydopamine; Vitiligo.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All animal studies were approved by the Animal Ethics and Welfare Committee of the First Affiliated Hospital of Wenzhou Medical University. Consent for publication: All authors have agreed to publish this manuscript. Competing interests: The authors declare no competing interests.
Figures






Similar articles
-
Oxidative stress-induced IL-15 trans-presentation in keratinocytes contributes to CD8+ T cells activation via JAK-STAT pathway in vitiligo.Free Radic Biol Med. 2019 Aug 1;139:80-91. doi: 10.1016/j.freeradbiomed.2019.05.011. Epub 2019 May 10. Free Radic Biol Med. 2019. PMID: 31078730
-
Janus Kinase Inhibitors in the Treatment of Vitiligo: A Review.Front Immunol. 2021 Nov 18;12:790125. doi: 10.3389/fimmu.2021.790125. eCollection 2021. Front Immunol. 2021. PMID: 34868078 Free PMC article. Review.
-
Janus kinase inhibitors and the changing landscape of vitiligo management: a scoping review.Int J Dermatol. 2024 Aug;63(8):1020-1035. doi: 10.1111/ijd.17157. Epub 2024 Apr 12. Int J Dermatol. 2024. PMID: 38610078
-
Pharmacological inhibition of demethylzeylasteral on JAK-STAT signaling ameliorates vitiligo.J Transl Med. 2023 Jul 4;21(1):434. doi: 10.1186/s12967-023-04293-2. J Transl Med. 2023. PMID: 37403086 Free PMC article.
-
Interferon-γ induces senescence in normal human melanocytes.PLoS One. 2014 Mar 28;9(3):e93232. doi: 10.1371/journal.pone.0093232. eCollection 2014. PLoS One. 2014. PMID: 24681574 Free PMC article.
Cited by
-
Leflunomide-Mediated Immunomodulation Inhibits Lesion Progression in a Vitiligo Mouse Model.Int J Mol Sci. 2025 Jul 15;26(14):6787. doi: 10.3390/ijms26146787. Int J Mol Sci. 2025. PMID: 40725033 Free PMC article.
-
Recent advances and perspectives of MicroNeedles for biomedical applications.Biophys Rev. 2025 Apr 24;17(3):909-928. doi: 10.1007/s12551-025-01317-7. eCollection 2025 Jun. Biophys Rev. 2025. PMID: 40727662 Free PMC article. Review.
References
-
- Akl J, Lee S, Ju HJ, Parisi R, Kim JY, Jeon JJ, Heo Y-W, Eleftheriadou V, Hamzavi I, Griffiths CEM, et al. Estimating the burden of vitiligo: a systematic review and modelling study. Lancet Public Health. 2024;9(6):e386–96. - PubMed
-
- Bastonini E, Kovacs D, Briganti S, Ottaviani M, D’Arino A, Migliano E, Pacifico A, Iacovelli PPM. Effects of pioglitazone on the differentiation and inflammation in vitiligo keratinocytes. J Eur Acad Dermatol Venereol. 2024;38(7):e573–5. - PubMed
-
- Mukhatayev ZL, Poole IC. Vitiligo: advances in pathophysiology research and treatment development. Trends Mol Med. 2024;30(9):844–62. - PubMed
-
- Renert-Yuval Y, Ezzedine K, Grimes P, Rosmarin D, Eichenfield LF, Castelo-Soccio L, Huang V, Desai SR, Walsh S, Silverberg JI, et al. Expert recommendations on Use of Topical therapeutics for Vitiligo in Pediatric, adolescent, and Young Adult patients. JAMA Dermatol. 2024;160(4):453–61. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

