α-Lipoic Acid Ameliorates Arsenic-Induced Lipid Disorders by Promoting Peroxisomal β-Oxidation and Reducing Lipophagy in Chicken Hepatocyte
- PMID: 39887668
- PMCID: PMC11923885
- DOI: 10.1002/advs.202413255
α-Lipoic Acid Ameliorates Arsenic-Induced Lipid Disorders by Promoting Peroxisomal β-Oxidation and Reducing Lipophagy in Chicken Hepatocyte
Abstract
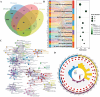
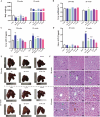
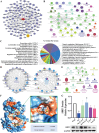
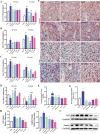
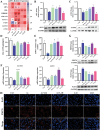
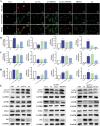
Liver disease poses a significant threat to global public health, with arsenic (As) recognized as a major environmental toxin contributing to liver injury. However, the specific mechanisms and the protective effects of α-lipoic acid (LA) remain unclear. Therefore, this study employs network toxicology and network pharmacology to comprehensively analyze the hepatotoxic mechanism of As and the hepatoprotective mechanism of LA, and further verifies the mechanisms of peroxisomal β-oxidation and lipophagy in the process. The network analysis results show that As induces liver damage mainly through autophagy, apoptosis, lipid metabolism, and oxidative stress, whereas LA exerts its hepatoprotective properties mainly by regulating lipid metabolism. Further verifications find that As inhibits SIRT1 expression, activates the P53 and Notch pathways, damages mitochondria, inhibits peroxisomal β-oxidation, increases lipid accumulation, and enhances lipophagy in the liver, while LA intervention alleviates As-induced lipid accumulation and enhances lipophagy by targeting SIRT1, ameliorating mitochondrial damage, enhancing peroxisomal β-oxidation, thereby alleviating As-induced liver damage. This study further clarifies the mechanism of As hepatotoxicity and provides a theoretical basis for LA as a potential hepatoprotective agent.
Keywords: alpha‐lipoic acid; arsenic; hepatotoxicity; lipophagy; peroxisomal β‐oxidation.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



References
-
- Harshad D., Sumeet K A., Juan Pablo A., Yvonne Ayerki N., Elisa P., Kamath P. S., J. Hepatol. 2023, 79, 516. - PubMed
-
- Lawal R. A., Hanotte O., Anim. Genet. 2021, 52, 385. - PubMed
-
- Wu J., Liang J., Björn L. O., Li J., Shu W., Wang Y., Sci. Total Environ. 2022, 802, 149796. - PubMed
-
- Podgorski J., Berg M., Science 2020, 368, 845. - PubMed
-
- Chen H., Zhang H., Wang X., Wu Y., Zhang Y., Chen S., Zhang W., Sun X., Zheng T., Xia W., Xu S., Li Y., Environ. Int. 2023, 174, 107918. - PubMed
MeSH terms
Substances
Grants and funding
- 2023YFD1801100/National Key Research and Development Program of China
- 32072934/National Natural Science Foundation of China
- 20210302124063/Fundamental Research Program of Shanxi Province
- 202403021211193/Fundamental Research Program of Shanxi Province
- 2022-101/Research Project Supported by Shanxi Scholarship Council of China
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous
