Synchronicity of pyramidal neurones in the neocortex dominates isoflurane-induced burst suppression in mice
- PMID: 39890488
- PMCID: PMC11947606
- DOI: 10.1016/j.bja.2024.10.052
Synchronicity of pyramidal neurones in the neocortex dominates isoflurane-induced burst suppression in mice
Abstract
Background: Anaesthesia-induced burst suppression signifies profound cerebral inactivation. Although considerable efforts have been directed towards elucidating the electroencephalographic manifestation of burst suppression, the neuronal underpinnings that give rise to isoflurane-induced burst suppression are unclear.
Methods: Electroencephalography combined with micro-endoscopic calcium imaging was used to investigate the neural mechanisms of isoflurane-induced burst suppression. Synchronous activities of pyramidal neurones in the auditory cortex and medial prefrontal cortex and inhibitory neurones in the auditory cortex (including parvalbumin [PV], somatostatin [SST], and vasoactive intestinal peptide [Vip]) and subcortical regions (including the medial geniculate body, locus coeruleus, and thalamic reticular nucleus) were recorded during isoflurane anaesthesia. In addition, the effects of chemogenetic manipulation inhibitory neurones in the auditory cortex on isoflurane-induced burst suppression were studied.
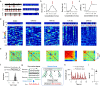
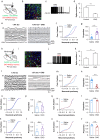
Results: Isoflurane-induced burst suppression was highly correlated with the synchronous activities of excitatory neurones in the cortex (∼65% positively and ∼20% negatively correlated neurones). Conversely, a minimal or absent correlation was observed with the neuronal synchrony of inhibitory interneurones and with neuronal activities within subcortical areas. Only activation or inhibition of PV neurones, but not SST or Vip neurones, decreased (P<0.0001) or increased (P<0.0001) isoflurane-induced neuronal synchrony.
Conclusions: Isoflurane-induced burst suppression might be primarily driven by the synchronous activities of excitatory pyramidal neurones in the cortex, which could be bidirectionally regulated by manipulating the activity of inhibitory PV interneurones. Our findings provide new insights into the neuronal mechanisms underlying burst suppression.
Keywords: auditory cortex; burst suppression; inhibitory neurones; isoflurane; micro-endoscopic calcium imaging; neuronal synchrony.
Copyright © 2025 British Journal of Anaesthesia. Published by Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interest The authors declare that they have no conflicts of interest.
Figures




Similar articles
-
The volatile anesthetic isoflurane differentially inhibits voltage-gated sodium channel currents between pyramidal and parvalbumin neurons in the prefrontal cortex.Front Neural Circuits. 2023 Jun 16;17:1185095. doi: 10.3389/fncir.2023.1185095. eCollection 2023. Front Neural Circuits. 2023. PMID: 37396397 Free PMC article.
-
Association between pyramidal neurone spiking in the medial prefrontal cortex and the sedative potency of volatile anaesthetics in mice.Br J Anaesth. 2025 Aug 26:S0007-0912(25)00492-1. doi: 10.1016/j.bja.2025.07.046. Online ahead of print. Br J Anaesth. 2025. PMID: 40866146
-
Cell-type-specific imaging of neurotransmission reveals a disrupted excitatory-inhibitory cortical network in isoflurane anaesthesia.EBioMedicine. 2021 Mar;65:103272. doi: 10.1016/j.ebiom.2021.103272. Epub 2021 Mar 7. EBioMedicine. 2021. PMID: 33691246 Free PMC article.
-
Distinct effects of volatile and intravenous anaesthetics on presynaptic calcium dynamics in mouse hippocampal GABAergic neurones.Br J Anaesth. 2022 Jun;128(6):1019-1028. doi: 10.1016/j.bja.2022.01.014. Epub 2022 Feb 11. Br J Anaesth. 2022. PMID: 35164969 Free PMC article.
-
Persistent gamma oscillations in superficial layers of rat auditory neocortex: experiment and model.J Physiol. 2005 Jan 1;562(Pt 1):3-8. doi: 10.1113/jphysiol.2004.074641. Epub 2004 Oct 15. J Physiol. 2005. PMID: 15489250 Free PMC article. Review.
Cited by
-
Solving the enigma of burst suppression.Br J Anaesth. 2025 Apr;134(4):900-902. doi: 10.1016/j.bja.2024.12.013. Epub 2025 Feb 21. Br J Anaesth. 2025. PMID: 40118581 Free PMC article.
-
Solving the enigma of burst suppression: when a seizure model is mistaken for burst suppression. Comment on Br J Anaesth 2025; 134: 900-2.Br J Anaesth. 2025 Sep;135(3):814-815. doi: 10.1016/j.bja.2025.06.001. Epub 2025 Jun 18. Br J Anaesth. 2025. PMID: 40537323 No abstract available.
References
-
- Akrawi W.P., Drummond J.C., Kalkman C.J., Patel P.M. A comparison of the electrophysiologic characteristics of EEG burst-suppression as produced by isoflurane, thiopental, etomidate, and propofol. J Neurosurg Anesthesiol. 1996;8:40–46. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

