Lymphatic-derived oxysterols promote anti-tumor immunity and response to immunotherapy in melanoma
- PMID: 39890772
- PMCID: PMC11893137
- DOI: 10.1038/s41467-025-55969-w
Lymphatic-derived oxysterols promote anti-tumor immunity and response to immunotherapy in melanoma
Abstract
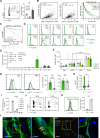
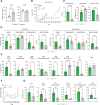
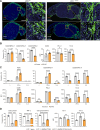
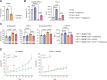
In melanoma, lymphangiogenesis correlates with metastasis and poor prognosis and promotes immunosuppression. However, it also potentiates immunotherapy by supporting immune cell trafficking. We show in a lymphangiogenic murine melanoma that lymphatic endothelial cells (LECs) upregulate the enzyme Ch25h, which catalyzes the formation of 25-hydroxycholesterol (25-HC) from cholesterol and plays important roles in lipid metabolism, gene regulation, and immune activation. We identify a role for LECs as a source of extracellular 25-HC in tumors inhibiting PPAR-γ in intra-tumoral macrophages and monocytes, preventing their immunosuppressive function and instead promoting their conversion into proinflammatory myeloid cells that support effector T cell functions. In human melanoma, LECs also upregulate Ch25h, and its expression correlates with the lymphatic vessel signature, infiltration of pro-inflammatory macrophages, better patient survival, and better response to immunotherapy. We identify here in mechanistic detail an important LEC function that supports anti-tumor immunity, which can be therapeutically exploited in combination with immunotherapy.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures



References
-
- Stacker, S. A. et al. Lymphangiogenesis and lymphatic vessel remodelling in cancer. Nat. Rev. Cancer14, 159–172 (2014). - PubMed
-
- Qian, C. N. et al. Preparing the “soil”: the primary tumor induces vasculature reorganization in the sentinel lymph node before the arrival of metastatic cancer cells. Cancer Res.66, 10365–10376 (2006). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

