Rescue of in vitro models of CSF1R-related adult-onset leukodystrophy by iluzanebart: mechanisms and therapeutic implications of TREM2 agonism
- PMID: 39891235
- PMCID: PMC11783791
- DOI: 10.1186/s12974-025-03346-1
Rescue of in vitro models of CSF1R-related adult-onset leukodystrophy by iluzanebart: mechanisms and therapeutic implications of TREM2 agonism
Abstract
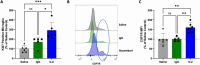
Microglia dysfunction is implicated in several neurodegenerative disorders, including a rare microgliopathy; CSF1R-related adult-onset leukoencephalopathy with axonal spheroids and pigmented glia (CSF1R-ALSP). CSF1R-ALSP is caused by heterozygous loss-of-function mutations in the colony stimulating factor 1 receptor (CSF1R) gene, which encodes a receptor required for the differentiation of myeloid cells, as well as for microglial survival and proliferation. Similar functions have also been ascribed to triggering receptor expressed on myeloid cells 2 (TREM2), which shares an analogous microglia enrichment profile and converging intracellular signaling pathway mediated by spleen associated tyrosine kinase (SYK) and phosphoinositide-3-kinase (PI3K). Iluzanebart is a human monoclonal IgG1, human TREM2 (hTREM2) agonist antibody under development for the treatment of CSF1R-ALSP. To explore the therapeutic hypothesis that loss of CSF1R signaling and related microglial hypofunction can be circumvented via activation of TREM2, we evaluated the potential of iluzanebart to compensate for CSF1R loss-of-function. Herein, we demonstrate that iluzanebart is a potent, dose-dependent, and specific activator of TREM2 signaling in human primary cells. Iluzanebart treatment rescued viability of human monocyte-derived macrophages (hMDM) and induced pluripotent stem cell-derived human microglia (iMGL) in multiple in vitro models of CSF1R-ALSP, including in induced pluripotent stem cell (iPSC) differentiated microglia carrying the heterozygous I794T mutation found in CSF1R-ALSP patients. Additionally, iluzanebart treatment in microglia modulated surface levels of CSF1R, resulting in increased receptor activation as measured by phosphorylation of CSF1R. Differentially expressed genes identified in the hippocampus of mice treated with iluzanebart were exemplary of TREM2 activation and were related to cell proliferation, regulation of inflammatory processes, and innate immune response pathways. Proliferation of microglia, changes in protein levels of specific chemokines identified by gene expression analysis, and increased CSF1R levels were also confirmed in vivo. These findings demonstrate that iluzanebart is a potent and selective TREM2 agonistic antibody, with pharmacology that supports the hypothesis that TREM2 activation can compensate for CSF1R dysfunction and its continued clinical development for individuals with CSF1R-ALSP.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: These studies followed national and institutional guidelines for humane animal treatment, with protocols reviewed and approved by the Charles River Laboratories Institutional Animal Care and Use Committee (IACUC). Competing interests: The authors declare no competing interests.
Figures










References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

