Tradeoffs in the energetic value of neuromodulation in a closed-loop neuromechanical system
- PMID: 39892775
- PMCID: PMC12007176
- DOI: 10.1016/j.jtbi.2025.112050
Tradeoffs in the energetic value of neuromodulation in a closed-loop neuromechanical system
Abstract
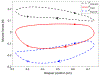
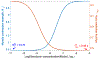
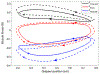
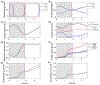
Rhythmic motor behaviors controlled by neuromechanical systems, consisting of central neural circuitry, biomechanics, and sensory feedback, show efficiency in energy expenditure. The biomechanical elements (e.g., muscles) are modulated by peripheral neuromodulation which may improve their strength and speed properties. However, there are relatively few studies on neuromodulatory control of muscle function and metabolic mechanical efficiency in neuromechanical systems. To investigate the role of neuromodulation on the system's mechanical efficiency, we consider a neuromuscular model of motor patterns for feeding in the marine mollusk Aplysia californica. By incorporating muscle energetics and neuromodulatory effects into the model, we demonstrate tradeoffs in the energy efficiency of Aplysia's rhythmic swallowing behavior as a function of the level of neuromodulation. A robust efficiency optimum arises from an intermediate level of neuromodulation, and excessive neuromodulation may be inefficient and disadvantageous to an animal's metabolism. This optimum emerges from physiological constraints imposed upon serotonergic modulation trajectories on the energy efficiency landscape. Our results may lead to experimentally testable hypotheses of the role of neuromodulation in rhythmic motor control.
Keywords: Central pattern generator; Closed-loop control; Energetics; Muscle; Neuromechanics; Neuromodulation.
Copyright © 2025 The Authors. Published by Elsevier Ltd.. All rights reserved.
Conflict of interest statement
Declaration of competing interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures
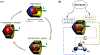

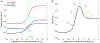



References
-
- Attwell D and Gibb A (2005). Neuroenergetics and the kinetic design of excitatory synapses. Nature Reviews Neuroscience, 6(11):841–849. - PubMed
-
- Attwell D and Laughlin SB (2001). An energy budget for signaling in the grey matter of the brain. Journal of Cerebral Blood Flow & Metabolism, 21(10):1133–1145. - PubMed
-
- Aubert X and Hill A (1956). Le couplage énergétique de la contraction musculaire. PhD thesis, UCL-Université Catholique de Louvain.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

