Stereotactic ablative radiotherapy (SABR) for pelvic nodal oligorecurrence in prostate cancer
- PMID: 39895963
- PMCID: PMC11785391
- DOI: 10.5603/rpor.101528
Stereotactic ablative radiotherapy (SABR) for pelvic nodal oligorecurrence in prostate cancer
Abstract
Background: This study evaluated the clinical outcomes of stereotactic ablative radiotherapy (SABR) in the treatment of oligometastatic pelvic node prostate cancer to delay androgen deprivation therapy (ADT).
Materials and methods: Pelvic lymph node metastases were identified by 11C-choline positron emission tomography (PET)-computed tomography (CT), and patients were not receiving ADT. SABR was administered using linear accelerators with intensity-modulated and image-guided radiotherapy, at a prescribed dose of 35 Gy in 5 fractions over 2 weeks. Response was assessed using Response Evaluation Criteria in Solid Tumours (RECIST) v1.1 criteria, and prostate-specific antigen (PSA) levels were monitored post-SABR. Toxicity and quality of life were assessed by the Common Terminology Criteria for Adverse Events Toxicity (CTCAE) v.5.0 and European Organisation for Research and Treatment of Cancer (EORTC) quality of life questionnaires QLQ-C30/QLQ-PR25, respectively. Kaplan-Meier and T-test were used for statistical analysis.
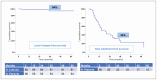
Results: Between June 2015 and November 2023, 56 patients with 85 lesions were treated at our institution. Median follow-up was 30 months [95% confidence interval (CI): 24-33.6]. Prostatectomy was the radical treatment in 85.7% of patients, and radiotherapy in 14.3%. Response rates were 67.1% for complete response, 27.4% for partial response, and 1.4% for stable disease. In-field progression was observed in only 3 lesions (3.5%). The median time to biochemical relapse post-SABR was 15 months (95% CI: 11.4-18.6). Three-year pelvic nodal and distant progression-free survival were 62.5% and 80%, respectively. There was a significant decrease in PSA levels after SABR compared to pretreatment levels (0.77 vs. 2.16 ng/mL respectively, p = 0.001). No grade ≥ 2 genitourinary or gastrointestinal toxicities. The median global health status score was 83.33 points at both time points analysed.
Conclusion: SABR can delay the ADT and provide excellent local control while preserving quality of life.
Keywords: oligometastases; oligorecurrence; pelvic lymph nodes; prostate cancer; stereotactic ablative radiotherapy.
© 2024 Greater Poland Cancer Centre.
Conflict of interest statement
Conflict of interest: None declared.
Figures
References
-
- Roach M, Hanks G, Thames H, et al. Defining biochemical failure following radiotherapy with or without hormonal therapy in men with clinically localized prostate cancer: recommendations of the RTOG-ASTRO Phoenix Consensus Conference. Int J Radiat Oncol Biol Phys. 2006;65(4):965–974. doi: 10.1016/j.ijrobp.2006.04.029. - DOI - PubMed
-
- Guckenberger M, Lievens Y, Bouma AB, et al. Characterisation and classification of oligometastatic disease: a European Society for Radiotherapy and Oncology and European Organisation for Research and Treatment of Cancer consensus recommendation. Lancet Oncol. 2020;21(1):e18–e28. doi: 10.1016/S1470-2045(19)30718-1. - DOI - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous