A randomised, double-masked, placebo-controlled trial evaluating the efficacy and safety of teprotumumab for active thyroid eye disease in Japanese patients
- PMID: 39896230
- PMCID: PMC11787687
- DOI: 10.1016/j.lanwpc.2025.101464
A randomised, double-masked, placebo-controlled trial evaluating the efficacy and safety of teprotumumab for active thyroid eye disease in Japanese patients
Erratum in
-
Corrigendum to "A randomised, double-masked, placebo-controlled trial evaluating the efficacy and safety of teprotumumab for active thyroid eye disease in Japanese patients" The Lancet Regional Health-Western Pacific, Volume 55 (2025), 101464.Lancet Reg Health West Pac. 2025 Jun 18;60:101607. doi: 10.1016/j.lanwpc.2025.101607. eCollection 2025 Jul. Lancet Reg Health West Pac. 2025. PMID: 40605970 Free PMC article.
Abstract
Background: Teprotumumab significantly improved proptosis and diplopia in patients with active, moderate-to-severe thyroid eye disease (TED) in previous North American and European studies. This is the first evaluation of efficacy and safety of teprotumumab for active, moderate-to-severe TED in Japanese patients.
Methods: This randomised, double-masked, placebo-controlled trial was conducted in 16 centres in Japan. Main inclusion criteria were as follows: age 20-80 years; Graves' disease, in a euthyroid or mild hypo/hyperthyroid state; clinical activity score (CAS) ≥3; moderate-to-severe TED; ≥3-mm increase in proptosis before TED onset and/or proptosis ≥18 mm at baseline; and TED duration ≤9 months. Patients were randomised (1:1, stratified by smoking status) to either teprotumumab or placebo. Patients received eight intravenous infusions, one every three weeks for 24 weeks. Patients, investigators, site personnel (except formulating pharmacists) were masked. Primary endpoint was proptosis responder rate (percentage of patients with ≥2-mm proptosis reduction from baseline) at week 24 in the intent-to-treat population. Adverse events were assessed in all patients. This trial was registered at Japan Registry for Clinical Trials (jRCT2031210453).
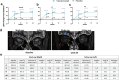
Findings: Fifty-four patients were randomised (teprotumumab, 27; placebo, 27) between February and November 2022. All patients completed the randomised period, although one teprotumumab patient and two placebo patients missed ≥2 doses. At week 24, the proportion of patients with proptosis response was higher in the teprotumumab group (89%, 24/27) compared with the placebo group (11%, 3/27), 95% confidence interval, 61-95; P<0.0001. Study drug-related adverse events (AEs) occurred in 14 patients (52%) in the teprotumumab group and two patients (7%) in the placebo group; hyperglycaemia-related events were reported in six (22%) and one patient (4%), and hearing impairment in four (15%) and one (4%) patient, respectively. Study drug-related serious AEs and deaths were not reported.
Interpretation: Teprotumumab significantly improved proptosis compared with placebo in Japanese patients with active TED. No study drug-related serious AEs were observed.
Funding: Horizon Therapeutics plc (now Amgen).
Keywords: Clinical activity score; Double-masked; Japanese; Placebo-controlled trial; Proptosis; Randomised; Teprotumumab; Thyroid eye disease.
© 2025 The Authors.
Conflict of interest statement
Horizon Therapeutics (now part of Amgen Inc.) sponsored and designed the trial with input from an investigator steering committee, contributed to collection, analysis, and interpretation of data, as well as writing, reviewing, and approval of the final version of the manuscript. Amgen Inc. (formerly Horizon Therapeutics) sponsored the study and funded the preparation of this manuscript. The authors’ institutions received funding from Amgen Inc. (formerly Horizon Therapeutics) for conducting the study. Y. Hiromatsu received grants from/has contracts with Chugai Pharmaceutical Co., Ltd and consulting fees from Amgen Inc (formerly Horizon Therapeutics). E. Ishikawa declares no additional conflicts. A. Kozaki received grants from/has contracts with Chugai Pharmaceutical Co., Ltd. Y. Takahashi declares no additional conflicts. M. Tanabe received payment or honoraria for lectures, presentations, speakers bureaus and consulting fees from Amgen Inc. (formerly Horizon Therapeutics); and received grants from/has contracts with Chugai Pharmaceutical Co., Ltd. K. Hayashi has received grants from/has contracts with Immunovant Inc. Y. Imagawa declares no additional conflicts. K. Kaneda declares no additional conflicts. M. Mimura received consulting fees and payment or honoraria for lectures, presentations, speakers bureaus from Amgen Inc. (formerly Horizon Therapeutics) and holds an unpaid fiduciary role at ASOPRS, JSOPRS and JSLPTD. T. Akamizu received consulting fees from Amgen Inc. (formerly Horizon Therapeutics). X. Dai and T. Hayashida are employees of Amgen Inc. (formerly of Horizon Therapeutics) and hold stock.
Figures

References
-
- Bahn R.S. Choice of therapy and criteria for assessing treatment outcome in thyroid-associated ophthalmopathy. N Engl J Med. 2010;362:726–738. - PubMed
-
- Kozaki A., Inoue R., Komoto N., et al. Proptosis in dysthyroid ophthalmopathy: a case series of 10,931 Japanese cases. Optom Vis Sci. 2010;87:200–204. - PubMed
-
- Chng C.L., Seah L.L., Khoo D.H. Ethnic differences in the clinical presentation of Graves' ophthalmopathy. Best Pract Res Clin Endocrinol Metab. 2012;26:249–258. - PubMed
LinkOut - more resources
Full Text Sources

