Radiomics in glioma: emerging trends and challenges
- PMID: 39901654
- PMCID: PMC11920724
- DOI: 10.1002/acn3.52306
Radiomics in glioma: emerging trends and challenges
Abstract
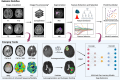
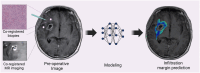
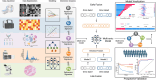
Radiomics is a promising neuroimaging technique for extracting and analyzing quantitative glioma features. This review discusses the application, emerging trends, and challenges associated with using radiomics in glioma. Integrating deep learning algorithms enhances various radiomics components, including image normalization, region of interest segmentation, feature extraction, feature selection, and model construction and can potentially improve model accuracy and performance. Moreover, investigating specific tumor habitats of glioblastomas aids in a better understanding of glioblastoma aggressiveness and the development of effective treatment strategies. Additionally, advanced imaging techniques, such as diffusion-weighted imaging, perfusion-weighted imaging, magnetic resonance spectroscopy, magnetic resonance fingerprinting, functional MRI, and positron emission tomography, can provide supplementary information for tumor characterization and classification. Furthermore, radiomics analysis helps understand the glioma immune microenvironment by predicting immune-related biomarkers and characterizing immune responses within tumors. Integrating multi-omics data, such as genomics, transcriptomics, proteomics, and pathomics, with radiomics, aids the understanding of the biological significance of the underlying radiomics features and improves the prediction of genetic mutations, prognosis, and treatment response in patients with glioma. Addressing challenges, such as model reproducibility, model generalizability, model interpretability, and multi-omics data integration, is crucial for the clinical translation of radiomics in glioma.
© 2025 The Author(s). Annals of Clinical and Translational Neurology published by Wiley Periodicals LLC on behalf of American Neurological Association.
Conflict of interest statement
There are no conflicts of interest to declare.
Figures

Similar articles
-
Applications of radiomics and machine learning for radiotherapy of malignant brain tumors.Strahlenther Onkol. 2020 Oct;196(10):856-867. doi: 10.1007/s00066-020-01626-8. Epub 2020 May 11. Strahlenther Onkol. 2020. PMID: 32394100 Free PMC article. Review.
-
Diffusion- and perfusion-weighted MRI radiomics model may predict isocitrate dehydrogenase (IDH) mutation and tumor aggressiveness in diffuse lower grade glioma.Eur Radiol. 2020 Apr;30(4):2142-2151. doi: 10.1007/s00330-019-06548-3. Epub 2019 Dec 11. Eur Radiol. 2020. PMID: 31828414
-
Comparative analysis of deep learning and radiomic signatures for overall survival prediction in recurrent high-grade glioma treated with immunotherapy.Cancer Imaging. 2025 Jan 21;25(1):5. doi: 10.1186/s40644-024-00818-0. Cancer Imaging. 2025. PMID: 39838503 Free PMC article.
-
Machine-Learning and Radiomics-Based Preoperative Prediction of Ki-67 Expression in Glioma Using MRI Data.Acad Radiol. 2024 Aug;31(8):3397-3405. doi: 10.1016/j.acra.2024.02.009. Epub 2024 Mar 7. Acad Radiol. 2024. PMID: 38458887
-
Machine learning applications to neuroimaging for glioma detection and classification: An artificial intelligence augmented systematic review.J Clin Neurosci. 2021 Jul;89:177-198. doi: 10.1016/j.jocn.2021.04.043. Epub 2021 May 13. J Clin Neurosci. 2021. PMID: 34119265
References
-
- Lapointe S, Perry A, Butowski NA. Primary brain tumours in adults. Lancet. 2018;392(10145):432‐446. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

