Long-term effects of dupilumab on chronic rhinosinusitis with nasal polyps: A step towards clinical remission
- PMID: 39902112
- PMCID: PMC11787525
- DOI: 10.1016/j.waojou.2024.101024
Long-term effects of dupilumab on chronic rhinosinusitis with nasal polyps: A step towards clinical remission
Abstract
Background and objectives: Clinical remission, defined as the absence of disease activity and symptoms, is an emerging goal in the management of chronic rhinosinusitis with nasal polyps (CRSwNP). This study aimed to evaluate the long-term effects of dupilumab on patients with CRSwNP, with or without asthma, and explore the potential for achieving clinical remission.
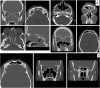
Methods: A two-year prospective study was conducted on 109 patients with CRSwNP, with or without asthma, who were eligible for dupilumab as an add-on therapy. Comprehensive assessments, including clinical, laboratory, and radiological evaluations, were performed before and after treatment. Clinical remission of CRSwNP was defined as 12 months of dupilumab treatment, no exacerbations requiring oral corticosteroids (OCS), no need for nasal sinus operation, no anosmia or hyposmia, a Sino-Nasal Outcome Test (SNOT-22) score under 20, and a Lund-Mackay score (LMS) below 10. For those with comorbid asthma, clinical remission was defined as an asthma control test (ACT) score of 19 or higher, no asthma exacerbations, and no need for OCS.
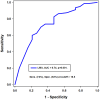
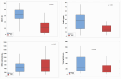
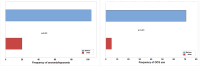
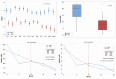
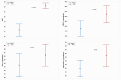
Results: Dupilumab significantly improved CRSwNP outcomes in both groups, including SNOT-22 scores, nasal polyp size (LMS), and anosmia/hyposmia. Comorbid asthma was highly prevalent (79.8%), and patients with asthma had significantly larger nasal polyps, both before and after dupilumab therapy, despite similar symptom improvement. Higher fractional exhaled nitric oxide (FeNO) and blood eosinophil count (BEC) levels, along with anosmia/hyposmia, predicted larger polyp size. Dupilumab also significantly improved asthma outcomes, increasing forced expiratory volume in 1 s (FEV1) and decreasing FeNO. Clinical remission was achieved in 11% of patients, with a slightly lower rate in those with asthma (7.3%).
Conclusion: Dupilumab treatment can achieve clinical remission in CRSwNP. However, comorbid asthma appears to reduce the likelihood of remission and is associated with larger nasal polyps, even with similar symptom improvement. Asthma may independently influence polyp development, potentially impacting long-term outcomes in CRSwNP.
Keywords: Chronic rhinosinusitis; Clinical remission; Comorbid asthma; Dupilumab; Lund-mackay score; Nasal polyps.
© 2025 The Authors.
Conflict of interest statement
All authors declare no conflict of interest. Each author has revised and approved the final version of the manuscript independently.
Figures
References
LinkOut - more resources
Full Text Sources

