Mitochondrial Peroxiredoxins and Monoamine Oxidase-A: Dynamic Regulators of ROS Signaling in Cardioprotection
- PMID: 39903882
- PMCID: PMC11835206
- DOI: 10.33549/physiolres.935513
Mitochondrial Peroxiredoxins and Monoamine Oxidase-A: Dynamic Regulators of ROS Signaling in Cardioprotection
Abstract
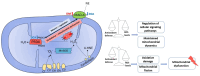
An excessive increase in reactive oxygen species (ROS) levels is one of the main causes of mitochondrial dysfunction. However, when ROS levels are maintained in balance with antioxidant mechanisms, ROS fulfill the role of signaling molecules and modulate various physiological processes. Recent advances in mitochondrial bioenergetics research have revealed a significant interplay between mitochondrial peroxiredoxins (PRDXs) and monoamine oxidase-A (MAO-A) in regulating ROS levels. Both proteins are associated with hydrogen peroxide (H2O2), MAO-A as a producer and PRDXs as the primary antioxidant scavengers of H2O2. This review focuses on the currently available knowledge on the function of these proteins and their interaction, highlighting their importance in regulating oxidative damage, apoptosis, and metabolic adaptation in the heart. PRDXs not only scavenge excess H2O2, but also act as regulatory proteins, play an active role in redox signaling, and maintain mitochondrial membrane integrity. Overexpression of MAO-A is associated with increased oxidative damage, leading to mitochondrial dysfunction and subsequent progression of cardiovascular diseases (CVD), including ischemia/reperfusion injury and heart failure. Considering the central role of oxidative damage in the pathogenesis of many CVD, targeting PRDXs activation and MAO-A inhibition may offer new therapeutic strategies aimed at improving cardiac function under conditions of pathological load related to oxidative damage. Keywords: Mitochondria, Peroxiredoxin, Monoamine oxidase-A, Reactive oxygen species, Cardioprotective signaling.
Conflict of interest statement
Figures
Similar articles
-
Interplay Between Mitochondrial Peroxiredoxins and ROS in Cancer Development and Progression.Int J Mol Sci. 2019 Sep 7;20(18):4407. doi: 10.3390/ijms20184407. Int J Mol Sci. 2019. PMID: 31500275 Free PMC article. Review.
-
Recent advances on the role of monoamine oxidases in cardiac pathophysiology.Basic Res Cardiol. 2023 Oct 4;118(1):41. doi: 10.1007/s00395-023-01012-2. Basic Res Cardiol. 2023. PMID: 37792081 Free PMC article.
-
Oxidative Stress by Monoamine Oxidase-A Impairs Transcription Factor EB Activation and Autophagosome Clearance, Leading to Cardiomyocyte Necrosis and Heart Failure.Antioxid Redox Signal. 2016 Jul 1;25(1):10-27. doi: 10.1089/ars.2015.6522. Epub 2016 Apr 22. Antioxid Redox Signal. 2016. PMID: 26959532
-
Monoamine oxidase B prompts mitochondrial and cardiac dysfunction in pressure overloaded hearts.Antioxid Redox Signal. 2014 Jan 10;20(2):267-80. doi: 10.1089/ars.2012.4616. Epub 2013 May 22. Antioxid Redox Signal. 2014. PMID: 23581564 Free PMC article.
-
Functions of Peroxiredoxins and Their Roles in Autoimmune Diseases.Antioxid Redox Signal. 2024 Feb;40(4-6):329-344. doi: 10.1089/ars.2022.0139. Epub 2023 Mar 27. Antioxid Redox Signal. 2024. PMID: 36738225 Review.
Cited by
-
Involvement of Oxidative Stress in Mitochondrial Abnormalities During the Development of Heart Disease.Biomedicines. 2025 May 29;13(6):1338. doi: 10.3390/biomedicines13061338. Biomedicines. 2025. PMID: 40564057 Free PMC article. Review.
References
-
- Trujillo M, Piacenza L, Radi R. Reactivity of mitochondrial peroxiredoxins with biological hydroperoxides. Redox Biochem Chem. 2023;5–6:100017. doi: 10.1016/j.rbc.2023.100017. - DOI
