Feasibility, Usability, and Pilot Efficacy Study of a Software-Enabled, Virtual Pulmonary Rehabilitation with Remote Therapeutic Monitoring
- PMID: 39906676
- PMCID: PMC11792638
- DOI: 10.2147/COPD.S484558
Feasibility, Usability, and Pilot Efficacy Study of a Software-Enabled, Virtual Pulmonary Rehabilitation with Remote Therapeutic Monitoring
Abstract
Objective: Fewer than 3% of adults with Chronic Obstructive Pulmonary Disease (COPD) attend in-person, center-based pulmonary rehabilitation (PR) despite demonstrated health benefits and reduction in mortality. This study evaluated the feasibility and usability of a novel home-based, virtual PR (V-PR) intervention compared to center-based PR (C-PR). The virtual PR intervention was supported by remote therapeutic monitoring (V-PR+RTM; Blue Marble Platform, Blue Marble Health, Altadena, CA). Additionally, we collected data on the 6-Minute Walk Test to explore the efficacy of the V-PR compared to C-PR.
Patients and methods: Adults with stable COPD referred for PR were recruited. The participants self-selected C-PR or V-PR and were provided a 6-8-week personalized exercise and COPD self-management educational program. In addition, weekly phone contacts with the V-PR group were made. Feasibility was measured using qualitative analysis of adherence, reasons for withdrawal, and self-reported barriers to using the software at home. Usability was measured with the System Usability Scale (SUS). Efficacy was evaluated with the 6 minute Walk Test (6MWT) and various functional performance and patient-centered health-related quality of life (HRQoL) questionnaires.
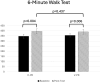
Results: Forty-eight participants were enrolled, and 40 (83.3%) completed the intervention, n=17 in the C-PR group and n=23 in the V-PR group. Four participants from each group withdrew due to reasons related to health issues (appendicitis, thrush, COVID, back pain) or the health status of their spouse, no-shows, and time constraints. Adherence to the exercise dose (3x/week) and educational offerings were >80% in both groups. Participants in the V-PR group scored the software as having high usability. In both groups, 6MWT distance improved significantly, as did scores on the CAT and SGRQ. No adverse events were reported in either group.
Conclusion: A software-enabled virtual PR program with remote therapeutic monitoring is feasible, usable, and effective. It could offer an alternative model that increases PR uptake for those unable or unwilling to attend in-person, center-based PR.
Keywords: COPD; chronic obstructive pulmonary disease; education; exercise training; pulmonary rehabilitation; remote therapeutic monitoring; self-management; telehealth; virtual.
Plain language summary
Pulmonary rehabilitation (PR) is a treatment program that improves breathlessness and quality of life for adults with lung diseases such as chronic obstructive pulmonary disease (COPD). Typically, PR is conducted in a clinic, which is often inaccessible for many adults with COPD; only 3% of Medicare-eligible patients with COPD participate in PR. Recently, new telehealth models of PR that do not require transport to a clinic have been shown to be as beneficial as clinic-based PR. This study measured the feasibility, usability, and effectiveness of a virtual home-based program supported by a remote therapeutic monitoring software platform and a weekly phone call from the clinician to the patient compared to traditional clinic-based PR. We found that both PR models were effective and that the virtual, home-based model supported by a remote monitoring software platform was feasible and usable.
© 2025 Flynn et al.
Conflict of interest statement
SF is the founder and CEO of Blue Marble Health. SF did not participate in the data collection of this study. CLM reports grants from Blue Marble Rehab, Inc, during the conduct of the study; research funding unrelated to this project from the American Lung Association, The Lang Family COPD Research Fund, Patient-Centered Outcomes Research Institute, AstraZeneca, and the National Institute on Aging/National Institutes of Health from R03AG082878-01 (GEMSSTAR award); grants from RofLumilast or Azithromycin to preveNt COPD Exacerbations (RELIANCE) (PCORI funded), A Phase III, Multicentre, Randomised, Double-blind, Chronic Dosing, Parallel Group, Placebo Controlled Study to Evaluate the Efficacy and Safety of Two Dose Regimens of MEDI3506 (AstraZeneca); personal fees from COPD Foundation, International Consulting Associates, Inc., Guidepoint, Physicians Education Resources, LLC, Wellinks, GlaxoSmithKline; travel reimbursement and honoraria from Respiratory Care Foundation, outside the submitted work. JO has received research funding from BI, TEVA, The Moore Foundation, The COPD Foundation, and NIH-Spiromics, as well as consulting fees from Theravance, Viatris, Chiesi Pharma, Astra Zeneca, Verona, Mylan, Genetech, and several law firms. NM reports personal fees from Inogen and Insprx. JD has received consulting fees from Medline Industries and educational fees from MedBridge. All remaining authors received funding from the NIH/NHLBI grant. The authors report no other conflicts of interest in this work.
Figures
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous