Usutu virus-induced meningoencephalitis in immunocompetent mice is characterized by the recruitment of mononuclear cells and a proinflammatory T helper 1 response
- PMID: 39907280
- PMCID: PMC11915786
- DOI: 10.1128/jvi.01724-24
Usutu virus-induced meningoencephalitis in immunocompetent mice is characterized by the recruitment of mononuclear cells and a proinflammatory T helper 1 response
Abstract
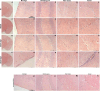
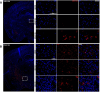
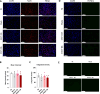
Usutu virus (USUV) is an arbovirus and has emerged as a potential cause of encephalitis in humans and other vertebrates. The increasing detection of USUV in mosquitoes and birds across Africa and Central Europe, along with the lack of specific treatments or vaccines for many encephalitic orthoflaviviruses, underscores the need for focused research. In this study, we developed a USUV infection model in immunocompetent C57BL/6 mice (8-12 weeks old) to characterize disease development and associated inflammatory mechanisms. Mice were intracranially infected with 104 PFU of USUV, leading to neurological symptoms such as hunched posture, paralysis, conjunctivitis, and eventual death by day 6 post-infection. Meningeal cell infiltration and microglia activation were most prevalent in mouse brains; however, neuronal loss was not observed at the peak of the disease, which coincided with increased viral load and leukocyte infiltration. The immune response in the brain was marked by the systematic recruitment and activation of macrophages, neutrophils, and T lymphocytes. A noticeable shift was seen in CD4+ T cells toward T helper 1 (Th1) polarization, which corroborates a massive increase in the expression of Th1-associated cytokines and chemokines at the peak of infection, indicative of an augmented proinflammatory state. Additionally, a rise in regulatory T cells was observed, peaking on day 6 post-infection. These findings highlight the dynamic nature of the host response to USUV infection, enhance our understanding of the disease pathogenesis, and address the scarcity of immunocompetent experimental models for the investigation of neglected emerging flaviviruses.IMPORTANCEMosquito-borne viruses, including USUV, are maintained in nature through complex cycles involving arthropod vectors and vertebrate hosts. A comprehensive understanding of USUV biology and host-pathogen interactions is crucial for developing effective treatments, which necessitates reliable experimental models (G. J. Sips, J. Wilschut, and J. M. Smit, Rev Med Virol 22:69-87, 2012, https://doi.org/10.1002/rmv.712; T. C. Pierson and M. S. Diamond, Nat Microbiol 5:796-812, 2020, https://doi.org/10.1038/s41564-020-0714-0). The establishment of a USUV infection model in immunocompetent adult mice brings new perspectives on the inflammatory component of viral encephalitis, which is difficult to study in mice lacking antiviral interferon responses. Moreover, USUV is an emerging viral disease lacking therapeutic and preventive measures. The interplay of USUV pathogenesis and the host's immune response indicates that lymphocytes and monocytes participate in USUV infection in this model and could be explored in search of treatments targeting immunopathogenic processes triggered by infection.
Keywords: Th1 response; Usutu virus; flavivirus; immunopathogenesis; leukocyte recruitment; mouse model; viral encephalitis.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
New Insights into the Susceptibility of Immunocompetent Mice to Usutu Virus.Viruses. 2020 Feb 8;12(2):189. doi: 10.3390/v12020189. Viruses. 2020. PMID: 32046265 Free PMC article.
-
Pathogenesis and shedding of Usutu virus in juvenile chickens.Emerg Microbes Infect. 2021 Dec;10(1):725-738. doi: 10.1080/22221751.2021.1908850. Emerg Microbes Infect. 2021. PMID: 33769213 Free PMC article.
-
Differential neurovirulence of Usutu virus lineages in mice and neuronal cells.J Neuroinflammation. 2021 Jan 6;18(1):11. doi: 10.1186/s12974-020-02060-4. J Neuroinflammation. 2021. PMID: 33407600 Free PMC article.
-
Usutu virus: an emerging flavivirus in Europe.Viruses. 2015 Jan 19;7(1):219-38. doi: 10.3390/v7010219. Viruses. 2015. PMID: 25606971 Free PMC article. Review.
-
Usutu virus: A new threat?Epidemiol Infect. 2019 Jan;147:e232. doi: 10.1017/S0950268819001213. Epidemiol Infect. 2019. PMID: 31364580 Free PMC article. Review.
Cited by
-
A central role for CCR2 in monocyte recruitment and blood-brain barrier disruption during Usutu virus encephalitis.J Neuroinflammation. 2025 Apr 16;22(1):107. doi: 10.1186/s12974-025-03435-1. J Neuroinflammation. 2025. PMID: 40241134 Free PMC article.
References
-
- Engel D, Jöst H, Wink M, Börstler J, Bosch S, Garigliany M-M, Jöst A, Czajka C, Lühken R, Ziegler U, Groschup MH, Pfeffer M, Becker N, Cadar D, Schmidt-Chanasit J. 2016. Reconstruction of the evolutionary history and dispersal of usutu virus, a neglected emerging arbovirus in europe and Africa. Mbio 7:e01938–15. doi:10.1128/mBio.01938-15 - DOI - PMC - PubMed
-
- Michel F, Sieg M, Fischer D, Keller M, Eiden M, Reuschel M, Schmidt V, Schwehn R, Rinder M, Urbaniak S, Müller K, Schmoock M, Lühken R, Wysocki P, Fast C, Lierz M, Korbel R, Vahlenkamp TW, Groschup MH, Ziegler U. 2019. Evidence for west nile virus and usutu virus infections in wild and resident birds in Germany, 2017 and 2018. Viruses 11:674. doi:10.3390/v11070674 - DOI - PMC - PubMed
MeSH terms
Substances
Supplementary concepts
Grants and funding
- 2021/05519-0, 2018/10990-1, 2016/00194-8, 2019/06459-1, 2020/04558-0, 2018/02594-9, 2019/20772-4/Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP)
- Ministério da Ciência, Tecnologia e Inovação (MCTI)
- 141253/2019-3/Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq)
- 01.20.0003.00/Financiadora de Estudos e Projetos (FINEP)
- 88887.351846/2019-00/Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES)
LinkOut - more resources
Full Text Sources
Research Materials

