Microbial Contamination of Percutaneous Endoscopic Gastrostomy Tube and Nasogastric Tube with Drug-Resistant Bacteria
- PMID: 39907997
- PMCID: PMC12136516
- DOI: 10.1159/000543972
Microbial Contamination of Percutaneous Endoscopic Gastrostomy Tube and Nasogastric Tube with Drug-Resistant Bacteria
Abstract
Introduction: Enteral nutrition is used in patients with stroke, head-and-neck or esophageal cancer surgery, or repeated aspiration pneumonia. Japanese enteral nutrition guidelines recommend percutaneous endoscopic gastrostomy tube (PEG) for long-term use of >4 weeks and nasogastric tube (NGT) for short-term use of <4 weeks. Catheters may be contaminated with microorganisms because enteral feeding products passed through them daily, but there are few reports on catheter contamination in Japan and no reports on the duration of catheter use. There are also reports that enteral feeding is a risk factor for the appearance of drug-resistant bacteria. Therefore, this study aimed to determine whether microorganisms could be isolated from enteral feeding catheters and determine the percentage of drug-resistant bacteria.
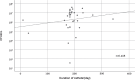
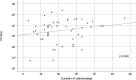
Methods: Forty-six PEGs and 59 NGTs were collected at Showa University Hospital and Showa University Fujigaoka Rehabilitation Hospital from May 2019 to March 2020. Microorganisms were cultured by incubating 20 mL pass/wash solution of sterile purified water on BHI agar medium (37°C) for 24-72 h. The strains were isolated and cultured, then frozen (80°C) and stored. Antimicrobial susceptibility was determined by broth microdilution method.
Results: Microorganisms were detected in 37 PEGs and 57 NGTs (p = 0.007). Bacteria were detected in 27 PEGs and 53 NGTs (p < 0.001), and yeasts were detected in 29 PEGs and 28 NGTs (p = 0.112). Drug-resistant bacteria were isolated from 19.6% (9 of 46) in PEGs and 23.7% (14 of 59) in NGTs.
Conclusions: PEGs and NGTs were contaminated with microorganisms, and drug-resistant bacteria were isolated. This study provides a rationale for future appropriate use in enteral feeding catheters.
Keywords: Bacteria; Drug-resistant bacteria; Enteral feeding; Nasogastric tube; Percutaneous endoscopic gastrostomy.
© 2025 The Author(s). Published by S. Karger AG, Basel.
Conflict of interest statement
The authors have no conflicts of interest to declare.
Figures
Similar articles
-
Transitioning From Nasogastric Feeding Tube to Gastrostomy Tube in Pediatric Patients: A Survey on Decision-Making and Practice.Nutr Clin Pract. 2021 Jun;36(3):654-664. doi: 10.1002/ncp.10603. Epub 2020 Dec 21. Nutr Clin Pract. 2021. PMID: 33347651
-
Microbial contamination of enteral feeding tubes occurring during nutritional treatment.JPEN J Parenter Enteral Nutr. 1992 Nov-Dec;16(6):552-7. doi: 10.1177/0148607192016006552. JPEN J Parenter Enteral Nutr. 1992. PMID: 1494212
-
[Enteral nutrition: nasogastric tube or percutaneous endoscopic gastrostomy?].Rev Med Suisse. 2012 Oct 17;8(358):1972-4, 1976-7. Rev Med Suisse. 2012. PMID: 23198651 French.
-
Enteral tube feeding in adults.J R Coll Physicians Edinb. 2015 Mar;45(1):49-54. doi: 10.4997/JRCPE.2015.112. J R Coll Physicians Edinb. 2015. PMID: 25874832 Review.
-
Use of gastrostomy and combined gastrojejunostomy tubes for enteral feeding.World J Surg. 1999 Jun;23(6):603-7. doi: 10.1007/pl00012354. World J Surg. 1999. PMID: 10227931 Review.
References
-
- JASPEN , Guidelines for venous enteral nutrition. 3rd ed. Tokyo: Shorinsha; 2016. Available from: https://minds.jcqhc.or.jp/common/wp-content/plugins/pdfjs-viewer-shortco...
-
- Okuma T, Nakamura M, Totake H, Fukunaga Y. Microbial contamination of enteral feeding formulas and diarrhea. Nutrition. 2000;16(9):719–22. - PubMed
-
- Sano M, Shindo Y, Takahashi K, Okumura J, Sakakibara T, Murakami Y, et al. . Risk factors for antibiotic resistance in hospital-acquired and ventilator-associated pneumonia. J Infect Chemother. 2022;28(6):745–52. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Medical