Perceptions of a Buruli ulcer controlled human infection model: How, who, and why?
- PMID: 39908304
- PMCID: PMC11819514
- DOI: 10.1371/journal.pntd.0012593
Perceptions of a Buruli ulcer controlled human infection model: How, who, and why?
Abstract
Background: Infection with Mycobacterium ulcerans causes slowly progressive skin lesions known as Buruli ulcer (BU). An M. ulcerans controlled human infection model (MuCHIM) is likely to accelerate our understanding of this otherwise neglected disease, and may be an efficient platform for testing vaccines and other interventions. The aim of this study was to understand perceptions of this model across a range of key stakeholders in an endemic Australian community setting.
Methods: We recruited young adults who live near an Australian BU endemic area but without a personal history of BU, clinicians involved in the management of BU, young adults with a personal history of a small, treated BU, and participants of any age with a demonstrated interest in public advocacy related to their personal BU lived experience. Participants reviewed an abridged version of the provisional protocol. A series of three focus groups were then conducted by video, and the transcribed text was analysed using reflexive thematic analysis to generate themes for exploration.
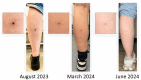
Results: Participants universally valued the outcomes that MuCHIM might deliver. The predominant theme was that informed consent required fully transparent communication with potential participants regarding what their participation would involve, how it would impact their lives, and both the expected outcome and 'worst-case scenario'. They also offered actionable recommendations on how best to communicate the tension between the expected outcome and the 'worst-case scenario' of disease associated with delayed diagnosis and comorbidity, as typically portrayed by the media. Participants recommended including images and testimonials from people who have had BU to support the conditions for informed consent. Focus groups also gave a clear sense of who they believed would volunteer for this type of research.
Conclusions: This study offers valuable guidance regarding the content and presentation of information to inform potential participants, with focus group participants suggesting a multimodal approach of communication, including lived experience testimonials and clinical images of the expected outcome. This information will inform development of materials for enrolment to adequately communicate risks and expectations to potential study participants.
Copyright: © 2025 Muhi et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- Muhi S, Osowicki J, O’Brien D, Johnson PDR, Pidot S, Doerflinger M, et al.. A human model of Buruli ulcer: the case for controlled human infection and considerations for selecting a Mycobacterium ulcerans challenge strain. PLoS Negl Trop Dis. 2023;17(6):e0011394. doi: 10.1371/journal.pntd.0011394 - DOI - PMC - PubMed
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

