Transmembrane voltage-gated nanopores controlled by electrically tunable in-pore chemistry
- PMID: 39910030
- PMCID: PMC11799347
- DOI: 10.1038/s41467-025-56052-0
Transmembrane voltage-gated nanopores controlled by electrically tunable in-pore chemistry
Abstract
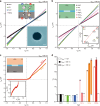
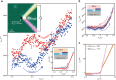
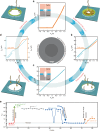
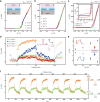
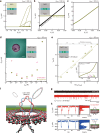
Gating is a fundamental process in ion channels configured to open and close in response to specific stimuli such as voltage across cell membranes thereby enabling the excitability of neurons. Here we report on voltage-gated solid-state nanopores by electrically tunable chemical reactions. We demonstrate repetitive precipitation and dissolution of metal phosphates in a pore through manipulations of cation flow by transmembrane voltage. Under negative voltages, precipitates grow to reduce ionic current by occluding the nanopore, while inverting the voltage polarity dissolves the phosphate compounds reopening the pore to ionic flux. Reversible actuation of these physicochemical processes creates a nanofluidic diode of rectification ratio exceeding 40000. The dynamic nature of the in-pore reactions also facilitates a memristor of sub-nanowatt power consumption. Leveraging chemical degrees of freedom, the present method may be useful for creating iontronic circuits of tunable characteristics toward neuromorphic systems.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures

References
Grants and funding
LinkOut - more resources
Full Text Sources

