Pulmonary aspergillosis in US Veterans with COVID-19: a nationwide, retrospective cohort study
- PMID: 39911504
- PMCID: PMC11795435
- DOI: 10.1017/ash.2024.476
Pulmonary aspergillosis in US Veterans with COVID-19: a nationwide, retrospective cohort study
Abstract
Background: COVID-associated pulmonary aspergillosis (CAPA) was described early in the pandemic as a complication of SARS-CoV-2. Data about incidence of aspergillosis and characteristics of affected patients after mid-2021 are limited.
Methods: A retrospective, nationwide cohort of US Veterans with SARS-CoV-2 from 1/1/2020 to 2/7/2024 was created. Potential cases of aspergillosis ≤12 weeks of a SARS-CoV-2 test were flagged electronically (based on testing results indicative of invasive fungal infection, antifungal therapy, and/or ICD-10 codes), followed by manual review to establish the clinical diagnosis of pulmonary aspergillosis. Incidence rates were calculated per 10,000 SARS-CoV-2 cases. Selected clinical characteristics included age >70, receipt of immune-compromising drugs, hematologic malignancy, chronic respiratory disease, vaccination status, and vaccine era. Multivariate logistic regression was used to estimate the independent effects of these variables via adjusted odds ratios (aOR).
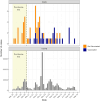
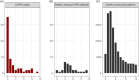
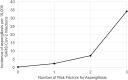
Results: Among 674,343 Veterans with SARS-CoV-2, 165 were electronically flagged for review. Of these, 66 were judged to be cases of aspergillosis. Incidence proportions ranged from 0.30/10,000 among patients with zero risk factors to 34/10,000 among those with ≥3 risk factors; rates were similar in the pre- and post-vaccination eras. The 90-day mortality among aspergillosis cases was 50%. In the multivariate analysis, immune suppression (aOR 6.47, CI 3.84-10.92), chronic respiratory disease (aOR 3.57, CI 2.10-6.14), and age >70 (aOR 2.78, CI 1.64-4.80) were associated with aspergillosis.
Conclusions: Patients with underlying risk factors for invasive aspergillosis continue to be at some risk despite SARS-CoV-2 immunization. Risk in patients without immune suppression or preexisting lung disease is very low.
© The Author(s) 2025.
Conflict of interest statement
All authors have no conflicts of interest to report.
Figures
Similar articles
-
COVID-19-associated pulmonary aspergillosis (CAPA) in hematological patients: Could antifungal prophylaxis be necessary? A nationwide study.J Infect Public Health. 2024 Jun;17(6):939-946. doi: 10.1016/j.jiph.2024.04.005. Epub 2024 Apr 10. J Infect Public Health. 2024. PMID: 38613930
-
Factors Associated With Severe COVID-19 Among Vaccinated Adults Treated in US Veterans Affairs Hospitals.JAMA Netw Open. 2022 Oct 3;5(10):e2240037. doi: 10.1001/jamanetworkopen.2022.40037. JAMA Netw Open. 2022. PMID: 36264571 Free PMC article.
-
COVID-19-associated pulmonary aspergillosis in patients with severe SARS-CoV-2 infection: A single-center observational study from Greece.Ann Thorac Med. 2023 Jul-Sep;18(3):116-123. doi: 10.4103/atm.atm_14_23. Epub 2023 May 16. Ann Thorac Med. 2023. PMID: 37663880 Free PMC article.
-
COVID-19-Associated Pulmonary Aspergillosis (CAPA).J Intensive Med. 2021 Aug 7;1(2):71-80. doi: 10.1016/j.jointm.2021.07.001. eCollection 2021 Oct. J Intensive Med. 2021. PMID: 36785564 Free PMC article. Review.
-
Risk factors for COVID-19-associated pulmonary aspergillosis: a systematic review and meta-analysis.Lancet Respir Med. 2024 Mar;12(3):207-216. doi: 10.1016/S2213-2600(23)00408-3. Epub 2024 Jan 4. Lancet Respir Med. 2024. PMID: 38185135
References
LinkOut - more resources
Full Text Sources
Miscellaneous
