Society for Cardiovascular Magnetic Resonance reference values ("normal values") in cardiovascular magnetic resonance: 2025 update
- PMID: 39914499
- PMCID: PMC12159681
- DOI: 10.1016/j.jocmr.2025.101853
Society for Cardiovascular Magnetic Resonance reference values ("normal values") in cardiovascular magnetic resonance: 2025 update
Abstract
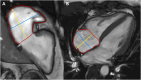
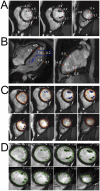
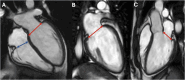
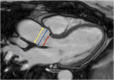
Quantitative assessment of morphological and functional cardiac parameters by cardiovascular magnetic resonance (CMR) is essential for research and routine clinical practice. Beyond established parameters of chamber size and function, tissue properties such as relaxation times play an increasing role. Normal reference ranges are required for interpretation of results obtained by quantitative CMR. Since the last publication of the "normal values review" in 2020 many new publications related to CMR reference values have been published, which were integrated in this update. The larger sample size provides greater statistical confidence in the estimates of upper and lower limits, and enables further partitioning, e.g., by age and ethnicity for several parameters. Previous topics were expanded by new sections.
Keywords: Cardiovascular magnetic resonance; Normal values; Reference range.
Copyright © 2025 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of competing interests David Bluemke reports a relationship with GE Healthcare that includes consulting or advisory. Co-author editor for the Journal of Cardiovascular Magnetic Resonance: Christopher François. The other authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Figures











References
-
- Horowitz G.L. Estimating reference intervals. Am J Clin Pathol. 2010;133(2):175–177. - PubMed
-
- Schulz-Menger J., Bluemke D.A., Bremerich J., Flamm S.D., Fogel M.A., Friedrich M.G., et al. Standardized image interpretation and post-processing in cardiovascular magnetic resonance-2020 update: Society for Cardiovascular Magnetic Resonance (SCMR): Board of Trustees Task Force on Standardized Post-Processing. J Cardiovasc Magn Reson. 2020;22(1) - PMC - PubMed
-
- R Core Team . R Foundation for Statistical Computing; Vienna, Austria: 2021. R: A language and environment for statistical computing.
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

