Intratumoral and peritumoral PET/CT-based radiomics for non-invasively and dynamically predicting immunotherapy response in NSCLC
- PMID: 39930148
- PMCID: PMC11920075
- DOI: 10.1038/s41416-025-02948-z
Intratumoral and peritumoral PET/CT-based radiomics for non-invasively and dynamically predicting immunotherapy response in NSCLC
Abstract
Background: We aimed to develop a machine learning model based on intratumoral and peritumoral 18F-FDG PET/CT radiomics to non-invasively and dynamically predict the response to immunotherapy in non-small cell lung cancer (NSCLC).
Methods: This retrospective study included 296 NSCLC patients, including a training cohort (N = 183), a testing cohort (N = 78), and a TCIA radiogenomic cohort (N = 35). The extreme gradient boosting algorithm was employed to develop the radiomic models.
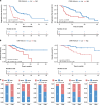
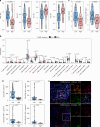
Results: The COMB-Radscore, which was developed by combining radiomic features from PET, CT, and PET/CT images, had the most satisfactory predictive performance with AUC (ROC) 0.894 and 0.819 in the training and testing cohorts, respectively. Survival analysis has demonstrated that COMB-Radscore is an independent prognostic factor for progression-free survival and overall survival. Moreover, COMB-Radscore demonstrates excellent dynamic predictive performance, with an AUC (ROC) of 0.857, enabling the earlier detection of potential disease progression in patients compared to radiological evaluation solely relying on tumor size. Further radiogenomic analysis showed that the COMB-Radscore was associated with infiltration abundance and functional status of CD8 + T cells.
Conclusions: The radiomic model holds promise as a precise, personalized, and dynamic decision support tool for the treatment of NSCLC patients.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: All methods and procedures performed in studies involving human participants were conducted according to the ethical standards of national research committee and the principles of the Declaration of Helsinki. This retrospective study was approved by the ethics committee of Nanfang Hospital of Southern Medical University (NFEC-2019-265). Patient informed consent was obtained for the use of pathological specimens, while the use of clinical data, laboratory test results, and 18F-FDG PET/CT waived the need for patient informed consent.
Figures




References
-
- Park K, Vansteenkiste J, Lee KH, Pentheroudakis G, Zhou C, Prabhash K, et al. Pan-Asian adapted ESMO Clinical Practice Guidelines for the management of patients with locally-advanced unresectable non-small-cell lung cancer: a KSMO-ESMO initiative endorsed by CSCO, ISMPO, JSMO, MOS, SSO and TOS. Ann Oncol. 2020;31:191–201. - DOI - PubMed
-
- Mok T, Wu YL, Kudaba I, Kowalski DM, Cho BC, Turna HZ, et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): a randomised, open-label, controlled, phase 3 trial. Lancet. 2019;393:1819–30. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

