Substance P receptor signaling contributes to host maladaptive responses during enteric bacterial infection
- PMID: 39937862
- PMCID: PMC11848390
- DOI: 10.1073/pnas.2415287122
Substance P receptor signaling contributes to host maladaptive responses during enteric bacterial infection
Abstract
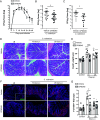
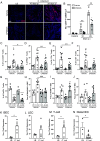
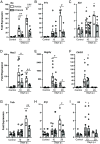
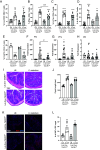
Immune responses in the intestine are intricately balanced to prevent pathogen entry without inducing immunopathology. The nervous system is well established to interface with the immune system to fine-tune immunity in various organ systems including the gastrointestinal tract. Specialized sensory neurons can detect bacteria, bacterial products, and the resulting inflammation, to coordinate the immune response in the gastrointestinal tract. These sensory neurons release peptide neurotransmitters such as Substance P (SP), to induce both neuronal signaling and localized responses in nonneuronal cells. With this in mind, we assessed the immunoregulatory roles of SP receptor signaling during enteric bacterial infection with the noninvasive pathogen Citrobacter rodentium. Pharmacological antagonism of the SP receptor significantly reduced bacterial burden and prevented colonic crypt hyperplasia. Mice with SP receptor signaling blockade had significantly reduced inflammation and recruitment of T cells in the colon. Reduced colonic T cell recruitment is due to reduced expression of adhesion molecules on colonic endothelial cells in SP receptor antagonist-treated mice. Using SP receptor T cell conditional knockout mice, we further confirmed SP receptor signaling enhanced select aspects of T cell responses. Our data demonstrate that SP receptor signaling can significantly reduce inflammation and prevent host-maladaptive responses without impinging upon host protection.
Keywords: Citrobacter rodentium; immunopathology; inflammation; mucosal Immunology; neuroimmunology.
Conflict of interest statement
Competing interests statement:The authors declare no competing interest.
Figures

Update of
-
Substance P receptor signaling contributes to host maladaptive responses during enteric bacterial infection.bioRxiv [Preprint]. 2024 Jun 27:2024.06.24.599421. doi: 10.1101/2024.06.24.599421. bioRxiv. 2024. Update in: Proc Natl Acad Sci U S A. 2025 Feb 18;122(7):e2415287122. doi: 10.1073/pnas.2415287122. PMID: 38979288 Free PMC article. Updated. Preprint.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

