Elucidating the Unique J-Shaped Protomer Structure of Amyloid-β(1-40) Fibril with Cryo-Electron Microscopy
- PMID: 39940945
- PMCID: PMC11817843
- DOI: 10.3390/ijms26031179
Elucidating the Unique J-Shaped Protomer Structure of Amyloid-β(1-40) Fibril with Cryo-Electron Microscopy
Abstract
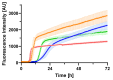
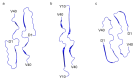
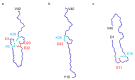
Although the structural diversity of amyloid-β (Aβ) fibrils plays a critical role in the pathology of Alzheimer's disease (AD), the mechanisms underlying this diversity remain poorly understood. In this study, we report the discovery of a novel J-shaped protomer structure of Aβ40 fibrils, resolved at 3.3 Å resolution using cryo-electron microscopy. Under controlled conditions (20 mM sodium phosphate buffer, pH 8.0) designed to emphasize intra-protomer interactions and slow fibril elongation, the J-shaped structure revealed distinct salt bridges (e.g., D1-K28, R5-E22) that stabilize the fibril core. These findings expand our understanding of the free energy landscape of fibril formation, shedding light on how specific environmental factors, such as pH and ionic strength, may influence fibril polymorphism. Importantly, the unique features of the J-shaped protomer provide insights into the structural basis of amyloid plaque diversity in AD and suggest potential therapeutic strategies targeting intra-protomer interactions. This study underscores the importance of fibril polymorphism in AD pathology and offers a foundation for future research into fibril-targeted therapies.
Keywords: Alzheimer’s disease; amyloid-β; cryo-electron microscopy; fibril.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

