Case report: PD-L1-targeted high-affinity natural killer cells and IL-15 superagonist N-803-based therapy extend overall survival of advanced metastatic pancreatic cancer patients
- PMID: 39944830
- PMCID: PMC11813753
- DOI: 10.3389/fonc.2025.1472714
Case report: PD-L1-targeted high-affinity natural killer cells and IL-15 superagonist N-803-based therapy extend overall survival of advanced metastatic pancreatic cancer patients
Abstract
Background: Metastatic pancreatic cancer (mPC) is an aggressive form of cancer with a poor prognosis and few therapeutic options after failure of the second-line treatment. Induction of immunogenic cell death (ICD) by use of relatively low-dose chemo- or radiation therapy, enhancement of immune responses by the IL-15 superagonist N-803 (Anktiva®), and targeting of programmed death receptor ligand 1 (PD-L1)-expressing cells may offer a therapeutic approach to refractory mPC with the potential to increase overall survival (OS).
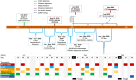
Methods: From late 2019 to 2021, single-patient Investigational New Drug (spIND) protocols for five mPC patients were designed and approved that generally comprised combined Abraxane (nab-paclitaxel) and gemcitabine therapy with experimental therapeutics N-803, PD-L1-targeted high-affinity natural killer (PD-L1 t-haNK) cells, and aldoxorubicin, a serum albumin-binding doxorubicin prodrug. Some patients also received stereotactic body radiation therapy (SBRT), cyclophosphamide, pembrolizumab, nivolumab, and/or experimental ETBX-051 (brachyury) and/or ETBX-061 (MUC1) vaccines. Duration of spIND treatment and responses, for some patients including imaging and carbohydrate antigen 19-9 (CA19-9) levels, and OS from initial diagnosis and the start of spIND therapy were assessed.
Findings: The line/duration of spIND therapy was, for patients 1 through 5, respectively, second line/6.4 months, sixth line/3.5 months, third line/25.4 months, third line/7.4 months, and fourth line/23.2 months. OS from the commencement of spIND therapy was 13, 4.8, 26.9, 9, and 23.2 months, and OS from diagnosis was 22, 21, 42, 13, and 33 months for patients 1 through 5, respectively.
Conclusions: The OS from the initiation of spIND for all patients exceeded the reported OS for the greater-than-second-line mPC patients and, for four of five patients, second-line therapy. The OS of 13, 26.9, and 23.2 months for three patients is particularly notable. The findings here support the ongoing clinical investigations of N-803 and PD-L1 t-haNK cells in combination therapy.
Keywords: 3rd line therapy; N-803; PD-L1 t-haNK cells; advanced metastatic pancreatic cancer; low dose chemotherapy; multi-modal; orchestrated.
Copyright © 2025 Seery, Sender, Jafari, Jones, Spilman, Reddy and Soon-Shiong.
Conflict of interest statement
TS, LS, FJ, PS, SR, and PS-S are employees or shareholders of ImmunityBio, Inc., which is developing N-803, PD-L1 t-haNK cells, and the described vaccines. PS-S is a shareholder in Celgene, acquired by the manufacturer of Abraxane, Bristol Myers Squibb. FJ was an original developer of ETBX vaccines and was employed by NantCell. The remaining author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



References
-
- NCI . Cancer stat facts: pancreatic cancer. In: National Cancer Institute (NCI) SEER Database (2021). Bethesda, MD: National Institutes of Health (NIH)/National Cancer Institute (NCI). https://seer.cancer.gov/statfacts/html/pancreas.html (Accessed July 19, 2023).
Publication types
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

