Effect of aging on pulmonary cellular responses during mechanical ventilation
- PMID: 39946196
- PMCID: PMC11949020
- DOI: 10.1172/jci.insight.185834
Effect of aging on pulmonary cellular responses during mechanical ventilation
Abstract
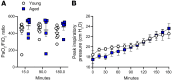
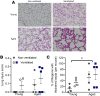
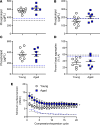
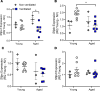
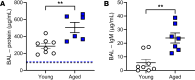
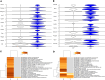
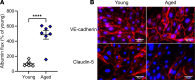
Acute respiratory distress syndrome (ARDS) results in substantial morbidity and mortality, especially in elderly people. Mechanical ventilation, a common supportive treatment for ARDS, is necessary for maintaining gas exchange but can also propagate injury. We hypothesized that aging leads to alterations in surfactant function, inflammatory signaling, and microvascular permeability within the lung during mechanical ventilation. Young and aged male mice were mechanically ventilated, and surfactant function, inflammation, and vascular permeability were assessed. Additionally, single-cell RNA-Seq was used to delineate cell-specific transcriptional changes. The results showed that, in aged mice, surfactant dysfunction and vascular permeability were significantly augmented, while inflammation was less pronounced. Differential gene expression and pathway analyses revealed that alveolar macrophages in aged mice showed a blunted inflammatory response, while aged endothelial cells exhibited altered cell-cell junction formation. In vitro functional analysis revealed that aged endothelial cells had an impaired ability to form a barrier. These results highlight the complex interplay between aging and mechanical ventilation, including an age-related predisposition to endothelial barrier dysfunction, due to altered cell-cell junction formation, and decreased inflammation, potentially due to immune exhaustion. It is concluded that age-related vascular changes may underlie the increased susceptibility to injury during mechanical ventilation in elderly patients.
Keywords: Aging; Endothelial cells; Macrophages; Pulmonary surfactants; Pulmonology; Vascular biology.
Figures



References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

