Long-Term Dynamics of CSF and Serum Neurofilament Light Chain in Adult Patients With 5q Spinal Muscular Atrophy Treated With Nusinersen
- PMID: 39946662
- PMCID: PMC11837849
- DOI: 10.1212/WNL.0000000000213371
Long-Term Dynamics of CSF and Serum Neurofilament Light Chain in Adult Patients With 5q Spinal Muscular Atrophy Treated With Nusinersen
Abstract
Background and objectives: The availability of disease-modifying therapies for 5q-associated spinal muscular atrophy (SMA) has heightened the need to identify suitable biomarkers. This study investigates neurofilament light chain (NfL) concentrations during long-term nusinersen treatment in adult SMA.
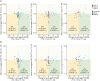
Methods: In a retrospective study of prospectively collected data, NfL concentrations in the CSF (cNfL) and serum (sNfL) were measured in patients with SMA from 8 German centers and in neurologic controls using a single-molecule array (Simoa) assay. NfL concentrations and clinical characteristics, including the clinical scores Hammersmith Functional Motor Scale Expanded (HFMSE), Revised Upper Limb Module (RULM), and Amyotrophic Lateral Sclerosis Functional Rating Scale-Revised (ALSFRS-R), were analyzed for defined treatment intervals (T1-T4 [loading phase until 4 months], T5-T8 [until 23 months], T9-T12 [until 37 months], and T13-T19 [until 60 months]). Linear mixed models with a random intercept were used to assess the changes in NfL levels during treatment, considering time and covariates as fixed effects.
Results: One hundred thirteen adult patients with SMA (median age 35, 46% female), with a treatment duration of maximum 60 months, and 52 controls were included. At baseline, NfL concentrations were significantly higher in SMA {cNfL median, 585 (interquartile range [IQR] 428-787) pg/mL; sNfL, 11 (IQR 8-14) pg/mL} than in controls (cNfL, 420 [IQR 323-662] pg/mL; sNfL, 8 [IQR 6-12] pg/mL) (cNfL, p = 0.021; sNfL, p = 0.030). Median differences for all clinical scores were the highest for T5-T8 compared with the loading phase (Δ HFMSE, 0.6 [IQR 0.1-1.4], p = 0.017; Δ RULM, 0.9 [IQR 0.4-1.3], p < 0.001; Δ ALSFRS-R, 0.7 [IQR 0.4-1.0], p < 0.001), but not for subsequent intervals. Longitudinal analysis revealed a significant decrease of NfL concentrations during each treatment interval compared with the loading phase (p < 0.05, respectively) except for sNfL in T13-T19. Even among patients with no measurable clinical improvement (Δ HFMSE ≤ 0), more than 50% showed declining cNfL and sNfL levels up to T13-T19.
Discussion: NfL decreased during nusinersen treatment, suggesting its potential as a pharmacodynamic response marker in adult SMA. However, in patients without detectable clinical improvement, our study cannot determine whether they represent a more sensitive outcome measure or are not clinically meaningful.
Conflict of interest statement
I. Cordts received speaker fees from Biogen and Hoffmann-La Roche outside of the submitted work and a research grant from Biogen. C. Fuetterer, A. Wachinger, R. von Heynitz, and T. Kessler report no disclosures relevant to the manuscript. M. Freigang received a speaker honorarium and nonfinancial support from Biogen outside the submitted work. A.L. Quinten, B. Bjelica, S. Brakemeier, and E. Hobbiebrunken report no disclosures relevant to the manuscript. T. Hagenacker received advisory board, speaker, and consultant honoraria from Biogen and Hoffmann-La Roche, Amylyx, Argenx, and Alexion and research support from Biogen, Hoffmann-La Roche, and Novartis outside of the submitted work. S. Petri received honoraria as a speaker/consultant from Biogen GmbH, Roche, Novartis, Teva, Cytokinetics Inc., Desitin, Italfarmaco, Amylyx, and Zambon and grants from DGM e.V, Federal Ministry of Education and Research, German Israeli Foundation for Scientific Research and Development, EU Joint Program for Neurodegenerative Disease Research, and Neurodegenerative Research, Inc., outside of the submitted work. J.C. Koch has received advisory board, speaker, and consultant honoraria from Biogen, Hoffmann-La Roche, Zambon, Merz, Ipsen, and AbbVie outside of the submitted work. A. Hahn reports no disclosures relevant to the manuscript. P. Lingor received honoraria for advisory boards and/or consultancies from Stada Pharm, Abbvie, Alexion, Bial, ITF Pharma, Desitin, Novartis, and Woolsey Pharma outside the scope of the submitted work. M. Deschauer received honoraria as a speaker/consultant from Biogen, Roche, and Sanofi outside the submitted work. R. Günther received personal honoraria as a speaker/consultant, personal fees from Biogen, Hofmann-La Roche, Zambon, and Sanofi ITF Pharma and research support from Biogen. All support was received outside the scope of the submitted work. M. Weiler received an advisory board and consultant honoraria from Biogen and Hoffmann-La Roche and speaker honoraria and travel support for conference attendance from Biogen outside the submitted work. M. Weiler is a member of the European Reference Network for Neuromuscular Diseases. B. Haller reports no disclosures relevant to the manuscript. E. Feneberg has applied for a US patent no. 17916203 outside of the submitted work and has received a research grant from Biogen. Go to
Figures



References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
