Neurovascular Decoupling Is Associated With Lobar Intracerebral Hemorrhages and White Matter Hyperintensities
- PMID: 39950450
- PMCID: PMC12074756
- DOI: 10.1161/JAHA.124.038819
Neurovascular Decoupling Is Associated With Lobar Intracerebral Hemorrhages and White Matter Hyperintensities
Abstract
Background: Neurovascular coupling is a fundamental aspect of brain function by regulating cerebral blood flow in response to regional neuronal activity. Increasing evidence suggest neurovascular decoupling occurs early in the progression of Alzheimer disease (AD), potentially reflecting early vascular damage. Therefore, understanding the relationship between neurovascular coupling and established vascular risk factors for AD is essential to gain deeper insights into the vascular mechanisms underlying AD.
Methods: This cross-sectional observational study investigated the association between neurovascular coupling and vascular risk factors for AD, specifically small vessel disease magnetic resonance imaging markers, cardiovascular risk factors, and the apolipoprotein E genotype. The cohort included 119 participants diagnosed with subjective cognitive impairment, mild cognitive impairment, and AD-related dementia, as well as individuals without cognitive complaints. Neurovascular coupling was measured by blood-oxygen-level-dependent functional magnetic resonance imaging amplitude in response to visual stimulation.
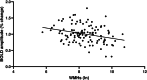
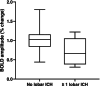
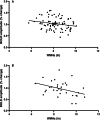
Results: Our findings revealed that decreased neurovascular coupling is linked to structural brain changes typically seen in small vessel disease; specifically we found an association between neurovascular coupling and white matter hyperintensities load (β=-0.199, P=0.030) and presence of lobar intracerebral hemorrhage (β=-0.228, P=0.011).
Conclusions: This raises the suggestion that a decreased neurovascular coupling in the disease process of AD is related to comorbid small vessel disease.
Keywords: Alzheimer disease; cerebral amyloid angiopathy; functional magnetic resonance imaging; neurovascular coupling; small vessel disease.
Conflict of interest statement
None.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

