LmCen-/- based vaccine is protective against canine visceral leishmaniasis following three natural exposures in Tunisia
- PMID: 39952958
- PMCID: PMC11828870
- DOI: 10.1038/s41541-025-01070-8
LmCen-/- based vaccine is protective against canine visceral leishmaniasis following three natural exposures in Tunisia
Abstract
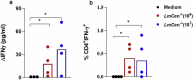
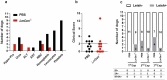
Dogs are the main reservoir host of Leishmania infantum, etiological agent of zoonotic visceral leishmaniasis (ZVL). An effective vaccine against Canine Visceral Leishmaniasis (CVL) will help the control and elimination of ZVL. In this study, we evaluated in dogs the safety, immunogenicity, and efficacy of a live attenuated Leishmania major Centrin gene-deleted (LmCen-/-) as a vaccine. Two doses (106 or 107) of LmCen-/- vaccine were administered intradermally in a prime-boost regimen. Both vaccine doses induced equally high level of IgG anti-Leishmania and exhibited strong antigen-specific cellular responses with IFN-γ production by CD4 + T cells one-month post-immunization. A second cohort of dogs was vaccinated with 106 LmCen-/- parasites one month prior to their transfer to a CVL endemic focus in Northern Tunisia for exposure to sand fly bites during three successive transmission seasons. Dogs were exposed to bite from naturally infected sandflies for 3-5 months per year. Our results showed that only 1/11 vaccinated dogs became PCR positive for Leishmania and developed clinical signs of CVL. In contrast, 4/11 unvaccinated dogs were tested PCR positive for Leishmania and displayed oligosymptomatic CVL, demonstrating that immunization with LmCen-/- vaccine confers long-term protection with an efficacy of 82.5% against CVL in natural transmission settings.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The FDA is currently the owner of two US patents that claim attenuated Leishmania species with the centrin gene deletion (US7,887,812 and US 8,877,213). All other authors declare no competing interests.
Figures


References
-
- (WHO), W.H.O. Leishmaniasis. 2022 [cited] Available from: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis.
-
- Alvar, J., Canavate, C., Molina, R., Moreno, J. & Nieto, J. Canine leishmaniasis. Adv. Parasitol.57, 1–88 (2004). - PubMed
-
- Koutinas, A. F. & Koutinas, C. K. Pathologic mechanisms underlying the clinical findings in canine leishmaniasis due to Leishmania infantum/chagasi. Vet. Pathol.51, 527–538 (2014). - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

