Identification of Meibomian gland stem cell populations and mechanisms of aging
- PMID: 39955307
- PMCID: PMC11830078
- DOI: 10.1038/s41467-025-56907-6
Identification of Meibomian gland stem cell populations and mechanisms of aging
Abstract
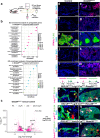
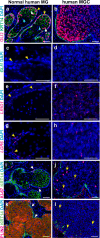
Meibomian glands secrete lipid-rich meibum, which prevents tear evaporation. Aging-related Meibomian gland shrinkage may result in part from stem cell exhaustion and is associated with evaporative dry eye disease, a common condition lacking effective treatment. The identities and niche of Meibomian gland stem cells and the signals controlling their activity are poorly defined. Using snRNA-seq, in vivo lineage tracing, ex vivo live imaging, and genetic studies in mice, we identify markers for stem cell populations that maintain distinct regions of the gland and uncover Hedgehog (Hh) signaling as a key regulator of stem cell proliferation. Consistent with this, we show that human Meibomian gland carcinoma exhibits increased Hh signaling. Aged glands display decreased Hh and EGF signaling, deficient innervation, and loss of collagen I in niche fibroblasts, indicating that alterations in both glandular epithelial cells and their surrounding microenvironment contribute to age-related degeneration. These findings suggest new approaches to treat aging-associated Meibomian gland loss.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures








Update of
-
Identification of Meibomian gland stem cell populations and mechanisms of aging.bioRxiv [Preprint]. 2024 Aug 10:2024.08.09.607015. doi: 10.1101/2024.08.09.607015. bioRxiv. 2024. Update in: Nat Commun. 2025 Feb 15;16(1):1663. doi: 10.1038/s41467-025-56907-6. PMID: 39149265 Free PMC article. Updated. Preprint.
References
-
- Obata, H. Anatomy and histopathology of human meibomian gland. Cornea21, S70–S74 (2002). - PubMed
-
- Wolff E., Bron A. J., Tripathi R. C., Tripathi B. J. Anatomy of the eye and orbit, 8th edn. Chapman & Hall Medical (1997).
MeSH terms
Substances
Grants and funding
- R37AR047709/U.S. Department of Health & Human Services | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- R01EY036135/U.S. Department of Health & Human Services | NIH | National Eye Institute (NEI)
- P30 AR069589/AR/NIAMS NIH HHS/United States
- R01 EY035337/EY/NEI NIH HHS/United States
- R01EY035337/U.S. Department of Health & Human Services | NIH | National Eye Institute (NEI)
- R37 AR047709/AR/NIAMS NIH HHS/United States
- R01 EY036135/EY/NEI NIH HHS/United States
- P30AR075043/U.S. Department of Health & Human Services | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- R01AR081322/U.S. Department of Health & Human Services | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- P30 CA046592/CA/NCI NIH HHS/United States
- P30 AR075043/AR/NIAMS NIH HHS/United States
- P30AR069589/U.S. Department of Health & Human Services | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- R01 AR081322/AR/NIAMS NIH HHS/United States
- R01AR065409/U.S. Department of Health & Human Services | NIH | National Institute of Arthritis and Musculoskeletal and Skin Diseases (NIAMS)
- P30 AR079200/AR/NIAMS NIH HHS/United States
- P30CA046592/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- R01 AR065409/AR/NIAMS NIH HHS/United States
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

