Measuring temperature in polyvinylpyrrolidone (PVP) solutions using MR spectroscopy
- PMID: 39960041
- PMCID: PMC12082762
- DOI: 10.1002/mp.17683
Measuring temperature in polyvinylpyrrolidone (PVP) solutions using MR spectroscopy
Abstract
Background: Polyvinylpyrrolidone (PVP) water solutions could be used for cross-site and cross-vendor validation of diffusion-related measurements. However, since water diffusivity varies as a function of temperature, knowing the temperature of the PVP solution at the time of the measurement is fundamental in accomplishing this task.
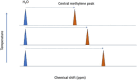
Purpose: MR spectroscopy (MRS) could provide absolute temperature measurements since the water peak moves relative to any stable peak as temperature changes. In this work, the PVP proton spectrum was investigated to see if any stable peaks would allow for temperature determination. Reproducibility and repeatability for three scanners from three vendors were also assessed.
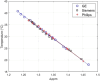
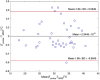
Methods: A spherical 17 cm container filled with 40% PVP w/w in distilled water was used for the experiments. A Point REsolved Spectroscopy Sequence (PRESS) with water suppression was employed on three 3T scanners from different vendors-GE, Siemens, and Philips. Frequency separation (in ppm) between peaks was measured in a voxel at the location of a fiber optic temperature probe and mapped to probe measured temperature. The center peak of the first methylene proton triplet closest to water peak was selected for analysis in jMRUI due to its ease of identification and echo time shift invariance. Shift in ppm of the central methylene peak proton was mapped against measured temperatures. Repeatability and reproducibility across the three scanners were determined at room temperature using 10 repeated PRESS scans. MRS established ppm shift versus temperature relationship was used to predict temperature in different PVP phantoms which were then compared against fiber optic probe measured temperature values.
Results: Several 1H peaks were identified on all scans of the PVP phantom. The water peak moved by ∼-0.01 ppm/°C on the three scanners relative to a central methylene peak. The maximum mean absolute temperature difference over a temperature range of 18-35°C between the three scanners was 0.16°C while the minimum was 0.057°C. Repeatability on each scanner was excellent (std range: 0.00-0.14°C) over 10 repeated PRESS scans. Reproducibility across the three scanners was also excellent with mean temperature difference between scanners ranging between 0.1 and 0.4°C. Temperature values from MRS were within prediction bounds on the three scanners for another in-house prepared 40% PVP phantom (maximum difference<0.3°C), while they were consistently overestimated for another 30% PVP phantom (<1°C) and underestimated for a CaliberMRI 40% PVP phantom (<2.8°C).
Conclusions: PVP solutions exhibit stable proton peaks, one of which was used for assessing the temperature of the solution using MR proton spectroscopy. These measurements are fast and feasible with standard sequences and postprocessing MRS software and provide fundamental information for calibration of diffusion MRI using PVP solutions.
Keywords: PVP phantom; PVP spectroscopy; polyvinylpyrrolidone; temperature mapping.
Published 2025. This article is a U.S. Government work and is in the public domain in the USA.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Pierpaoli C. Artifacts in diffusion MRI. In: Jones D, ed. Diffusion MRI: Theory, Methods, and Applications. Oxford University Press; 2012.
-
- Fukuzaki M, Alexander AL, Goodrich C, et al. The ability of line scan diffusion imaging method — comparison with echo planar diffusion imaging. Proc ISMRM. 1999:1833.
-
- Horkay F, Pierpaoli C, Basser PJ. Inventors; Dept of Health and Human Services, USA, assignee. Phantom for diffusion MRI imaging . US patent 9603546, 2017.
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials

