Time-resolved X-ray solution scattering unveils the events leading to hemoglobin heme capture by staphylococcal IsdB
- PMID: 39962089
- PMCID: PMC11832919
- DOI: 10.1038/s41467-024-54949-w
Time-resolved X-ray solution scattering unveils the events leading to hemoglobin heme capture by staphylococcal IsdB
Abstract
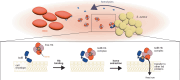
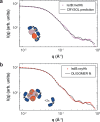
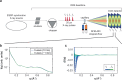
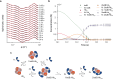
Infections caused by Staphylococcus aureus depend on its ability to acquire nutrients. One essential nutrient is iron, which is obtained from the heme of the human host hemoglobin (Hb) through a protein machinery called Iron-regulated surface determinant (Isd) system. IsdB is the protein in charge of heme extraction from Hb, which is the first step of the chain of events leading to iron transfer to the bacterium cell interior. In order to elucidate the molecular events leading from the formation of the initial IsdB:Hb complex to heme extraction, we use time-resolved X-ray solution scattering (TR-XSS) in combination with rapid mixing triggering. We succeed in defining the stoichiometry of IsdB:Hb binding and in describing the kinetics of the subsequent structural changes. The presented approach is potentially applicable to unveil the complex kinetic pathways generated by protein-protein interaction in different biological systems.
© 2024. The Author(s).
Conflict of interest statement
Competing interests: The authors declare no competing interests.
Figures

References
MeSH terms
Substances
Grants and funding
- IH-LS-3074 and LS-2807/European Synchrotron Radiation Facility (ESRF)
- PRIN 2020AE3LTA/Ministero dell'Istruzione, dell'Università e della Ricerca (Ministry of Education, University and Research)
- PRIN 2022RCP52Y/Ministero dell'Istruzione, dell'Università e della Ricerca (Ministry of Education, University and Research)
- Scientific Equipment Fund 2018/Università degli Studi di Parma (University of Parma)
LinkOut - more resources
Full Text Sources

