Nitric oxide inhibits ten-eleven translocation DNA demethylases to regulate 5mC and 5hmC across the genome
- PMID: 39966373
- PMCID: PMC11836389
- DOI: 10.1038/s41467-025-56928-1
Nitric oxide inhibits ten-eleven translocation DNA demethylases to regulate 5mC and 5hmC across the genome
Abstract
DNA methylation at cytosine bases (5-methylcytosine, 5mC) is a heritable epigenetic mark regulating gene expression. While enzymes that metabolize 5mC are well-characterized, endogenous signaling molecules that regulate DNA methylation machinery have not been described. We report that physiological nitric oxide (NO) concentrations reversibly inhibit the DNA demethylases TET and ALKBH2 by binding to the mononuclear non-heme iron atom forming a dinitrosyliron complex (DNIC) and preventing cosubstrates from binding. In cancer cells treated with exogenous NO, or endogenously synthesizing NO, 5mC and 5-hydroxymethylcytosine (5hmC) increase, with no changes in DNA methyltransferase activity. 5mC is also significantly increased in NO-producing patient-derived xenograft tumors from mice. Genome-wide methylome analysis of cells chronically treated with NO (10 days) shows enrichment of 5mC and 5hmC at gene-regulatory loci, correlating with altered expression of NO-regulated tumor-associated genes. Regulation of DNA methylation is distinctly different from canonical NO signaling and represents a unique epigenetic role for NO.
© 2025. The Author(s).
Conflict of interest statement
Competing interests: J.C. is the sole inventor on patent application no. 10420838 entitled: Methods for treating cancer using iNOS-inhibitory compositions, held by Houston Methodist Hospital. The other authors declare no competing interests.
Figures

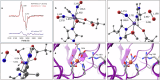


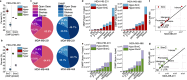

Update of
-
Nitric oxide inhibits ten-eleven translocation DNA demethylases to regulate 5mC and 5hmC across the genome.Res Sq [Preprint]. 2024 Apr 3:rs.3.rs-4131804. doi: 10.21203/rs.3.rs-4131804/v1. Res Sq. 2024. Update in: Nat Commun. 2025 Feb 18;16(1):1732. doi: 10.1038/s41467-025-56928-1. PMID: 38645113 Free PMC article. Updated. Preprint.
References
-
- Ferguson-Smith, A. C. Genomic imprinting: the emergence of an epigenetic paradigm. Nat. Rev. Genet.12, 565–575 (2011). - PubMed
-
- Illingworth, R. S. & Bird, A. P. CpG islands-‘a rough guide’. FEBS Lett.583, 1713–1720 (2009). - PubMed
-
- Loda, A., Collombet, S. & Heard, E. Gene regulation in time and space during X-chromosome inactivation. Nat. Rev. Mol. Cell Biol.23, 231–249 (2022). - PubMed
-
- Jones, P. A. Functions of DNA methylation: islands, start sites, gene bodies and beyond. Nat. Rev. Genet.13, 484–492 (2012). - PubMed
MeSH terms
Substances
Grants and funding
- R01 CA284315/CA/NCI NIH HHS/United States
- R01 CA217809/CA/NCI NIH HHS/United States
- CHE-2204114/National Science Foundation (NSF)
- R01 GM155946/GM/NIGMS NIH HHS/United States
- CA217809/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- R01GM155946/U.S. Department of Health & Human Services | NIH | National Institute of General Medical Sciences (NIGMS)
- R01 GM111097/GM/NIGMS NIH HHS/United States
- R01GM111097/U.S. Department of Health & Human Services | National Institutes of Health (NIH)
- U01 CA268813/CA/NCI NIH HHS/United States
- 17/CDA/4638/Science Foundation Ireland (SFI)
- U54 CA143924/CA/NCI NIH HHS/United States
- R01CA284315/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- R01GM130752/U.S. Department of Health & Human Services | National Institutes of Health (NIH)
- R01 HL170286/HL/NHLBI NIH HHS/United States
- U01CA268813/U.S. Department of Health & Human Services | NIH | National Cancer Institute (NCI)
- OAC-2117247/National Science Foundation (NSF)
- R01 GM130752/GM/NIGMS NIH HHS/United States
LinkOut - more resources
Full Text Sources

